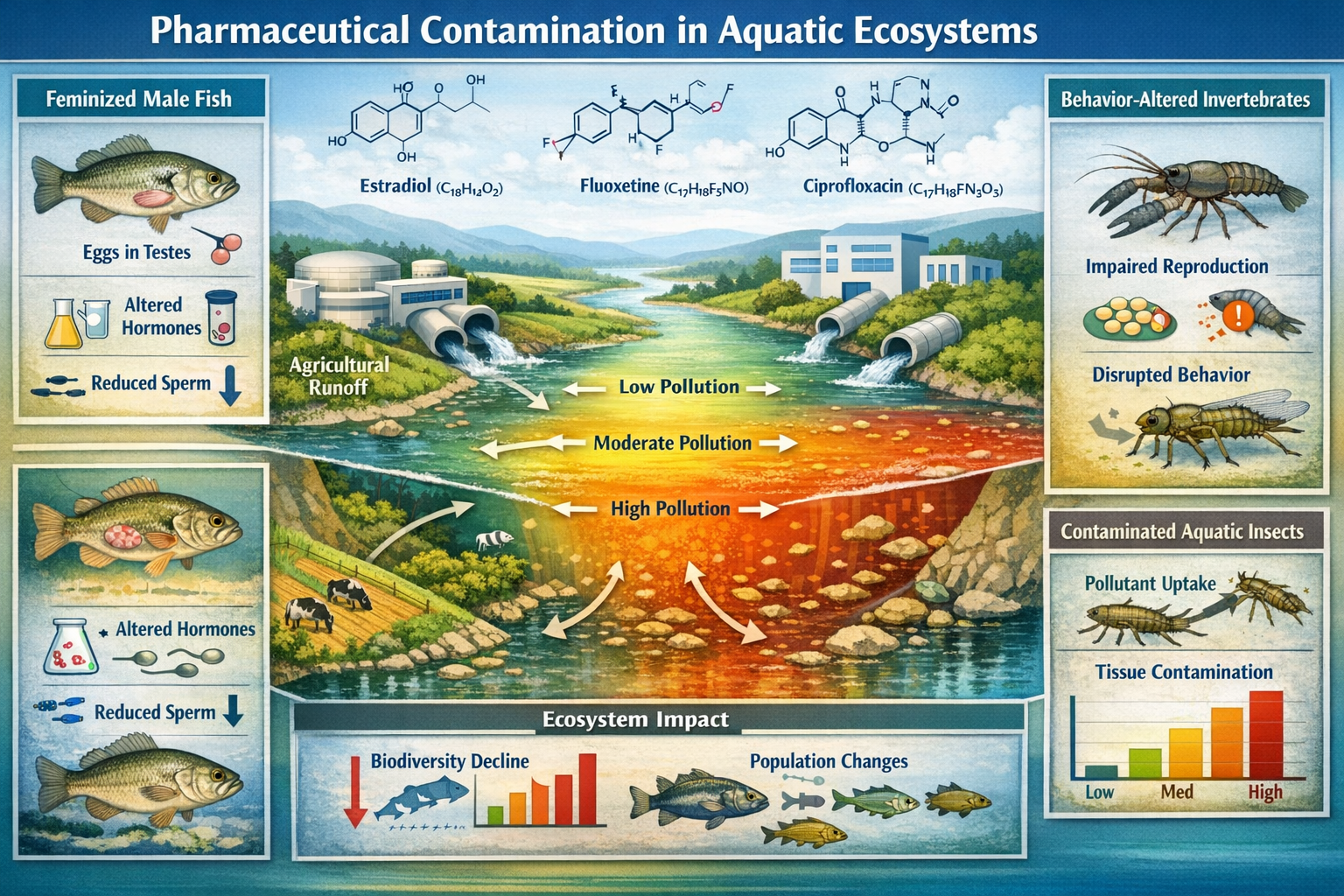
Estrogens from contraceptive pills are feminizing male fish in English rivers right now, reducing their breeding capacity and threatening population stability in protected waterways [3]. This stark reality represents just one dimension of pharmaceutical pollution's impact on aquatic biodiversity—a challenge that has escalated dramatically as of 2026, with contaminants now documented across national parks and biodiversity hotspots throughout the UK. For developers, landowners, and ecology professionals navigating Biodiversity Net Gain (BNG) requirements, understanding Pharmaceutical Pollutants in Biodiversity Surveys: eDNA Detection Strategies for Aquatic BNG Baselines has become essential for accurate environmental assessment and regulatory compliance.
The integration of pharmaceutical metabolite screening into standard biodiversity surveys represents a paradigm shift in how we establish aquatic baselines. Traditional survey methods often miss the invisible chemical stressors that fundamentally alter ecosystem function, even when species lists appear superficially healthy. Environmental DNA (eDNA) detection technologies now offer ecology surveyors powerful tools to simultaneously assess biological diversity and detect pharmaceutical contamination signatures—creating comprehensive baseline data that meets 2026 compliance standards while revealing hidden environmental risks.

Key Takeaways
- Pharmaceutical pollutants alter aquatic biodiversity through hormone disruption, behavioral changes, and ecosystem-wide impacts that traditional surveys often miss
- eDNA metabarcoding enables simultaneous species detection and pharmaceutical impact assessment, providing cost-effective baseline data for BNG compliance
- Integrated monitoring protocols combine broad metabarcoding surveys with targeted qPCR testing for specific pharmaceutical metabolites and indicator species
- 2026 compliance frameworks increasingly require pharmaceutical screening in aquatic BNG baselines, particularly for sites near wastewater discharge points
- Multi-functional biomonitoring approaches track pollution sources, contamination levels, ecological impacts, and bioindicator community composition in single sampling events
Understanding Pharmaceutical Pollution in Aquatic Ecosystems
The Scale and Sources of Pharmaceutical Contamination
Pharmaceutical pollution has emerged as a pervasive environmental challenge affecting waterways across England and globally. These contaminants enter aquatic systems through multiple pathways: wastewater treatment plant discharges, agricultural runoff containing veterinary pharmaceuticals, hospital effluent, and improper medication disposal [3]. Unlike point-source industrial pollutants, pharmaceutical contamination creates diffuse, persistent exposure scenarios that affect entire watershed systems.
Common pharmaceutical pollutants detected in UK waterways include:
- 💊 Hormone disruptors (contraceptive estrogens, hormone replacement therapies)
- 🧠 Psychoactive compounds (antidepressants, anxiolytics, stimulants)
- 💉 Antibiotics (human and veterinary formulations)
- 💓 Cardiovascular medications (beta-blockers, blood pressure regulators)
- 🩺 Anti-inflammatory drugs (ibuprofen, diclofenac, aspirin)
The biological activity of these compounds—designed specifically to affect physiological processes—creates unique ecological risks. Even at trace concentrations measured in nanograms per liter, pharmaceuticals can trigger measurable biological responses in non-target aquatic organisms [1].
Ecological Impacts on Aquatic Biodiversity
Recent research has documented alarming ecological consequences of pharmaceutical exposure in aquatic systems. Antidepressants alter fish behavior toward increased risk-taking, making treated populations more susceptible to predation and disrupting normal ecological hierarchies [3]. This behavioral modification cascades through food webs, affecting predator-prey dynamics and competitive interactions.
The impacts extend beyond direct aquatic exposure. Studies published in February 2026 revealed that pharmaceutical contaminants accumulate in aquatic insects at various life-cycle stages and transfer to terrestrial ecosystems, exposing riparian predators including birds, bats, and spiders to bioactive compounds via insect emergence [6]. This cross-ecosystem contamination pathway means pharmaceutical pollution affects biodiversity far beyond the water's edge—a critical consideration for comprehensive BNG assessments.
"Pharmaceutical pollution represents an invisible stressor that fundamentally alters ecosystem function, even when species presence appears normal through traditional survey methods."
For developers working on biodiversity net gain projects, understanding these pollution dynamics is essential. Baseline surveys that fail to account for pharmaceutical contamination may significantly overestimate habitat quality and ecological functionality, leading to inadequate mitigation measures and compliance failures.
Pharmaceutical Pollutants in Biodiversity Surveys: eDNA Detection Strategies for Aquatic BNG Baselines
What is Environmental DNA and How Does It Work?
Environmental DNA (eDNA) captures genetic material shed by organisms into water, soil, or air, enabling species detection without direct observation [4]. In aquatic environments, organisms continuously release DNA through skin cells, mucus, feces, gametes, and decomposing tissue. This genetic material persists in water for days to weeks, creating a molecular record of biological presence.
The eDNA detection process involves:
- Sample collection – Water samples collected using sterile filtration equipment
- DNA extraction – Genetic material isolated from filtered particulates
- Amplification – Target DNA sequences multiplied using PCR technology
- Sequencing – Genetic barcodes identified through metabarcoding or species-specific qPCR
- Bioinformatics analysis – Sequences matched to reference databases for species identification
This non-invasive approach revolutionizes biodiversity surveying by detecting rare, cryptic, or elusive species that traditional methods miss. Rapid eDNA surveys have detected 400 vertebrate species across 30,000 km² in western China within just 56 calendar days, including dozens of IUCN-listed species [4]—demonstrating the scalability and speed essential for comprehensive BNG baseline assessments.
Multi-Functional Biomonitoring for Pharmaceutical Pollution
eDNA metabarcoding provides multi-functional biomonitoring capabilities specifically suited to pharmaceutical pollution assessment [1]. This integrated approach enables:
- ✅ Pollution status assessment – Baseline contamination levels established through bioindicator presence/absence
- 🔍 Source identification – Upstream vs. downstream sampling reveals contamination entry points
- 📊 Temporal tracking – Repeated sampling monitors contamination level changes over time
- 🌿 Ecological impact assessment – Sensitive species loss indicates functional ecosystem degradation
- 🎯 Cleanup prioritization – Hotspot identification guides remediation resource allocation
- 🦠 Bioindicator community composition – Species assemblage shifts reveal pollution stress
This comprehensive monitoring framework addresses a critical gap in traditional biodiversity net gain assessments. Standard habitat condition assessments evaluate physical structure and dominant species but often miss chemical stressors that undermine ecological function. Pharmaceutical contamination may allow pollution-tolerant generalist species to persist while eliminating sensitive specialists—creating the illusion of biodiversity that masks functional ecosystem collapse.
Cost-Effectiveness and Scalability
Economic considerations drive eDNA adoption for large-scale BNG baseline surveys. Species-specific qPCR testing costs £200-500 per sample while metabarcoding detects 100+ species per sample [2], offering substantial cost advantages for comprehensive biodiversity assessment. This efficiency proves particularly valuable for developers managing multiple sites or extensive linear infrastructure projects requiring numerous sampling locations.
The scalability of eDNA surveys supports national biodiversity reporting targets and enables baseline/counterfactual assessments for restoration projects [4]. For landowners considering selling biodiversity units, robust eDNA baseline data strengthens habitat banking proposals by demonstrating both current ecological value and contamination-free conditions.
Implementing eDNA Protocols for 2026 BNG Compliance
Integrated Monitoring Framework
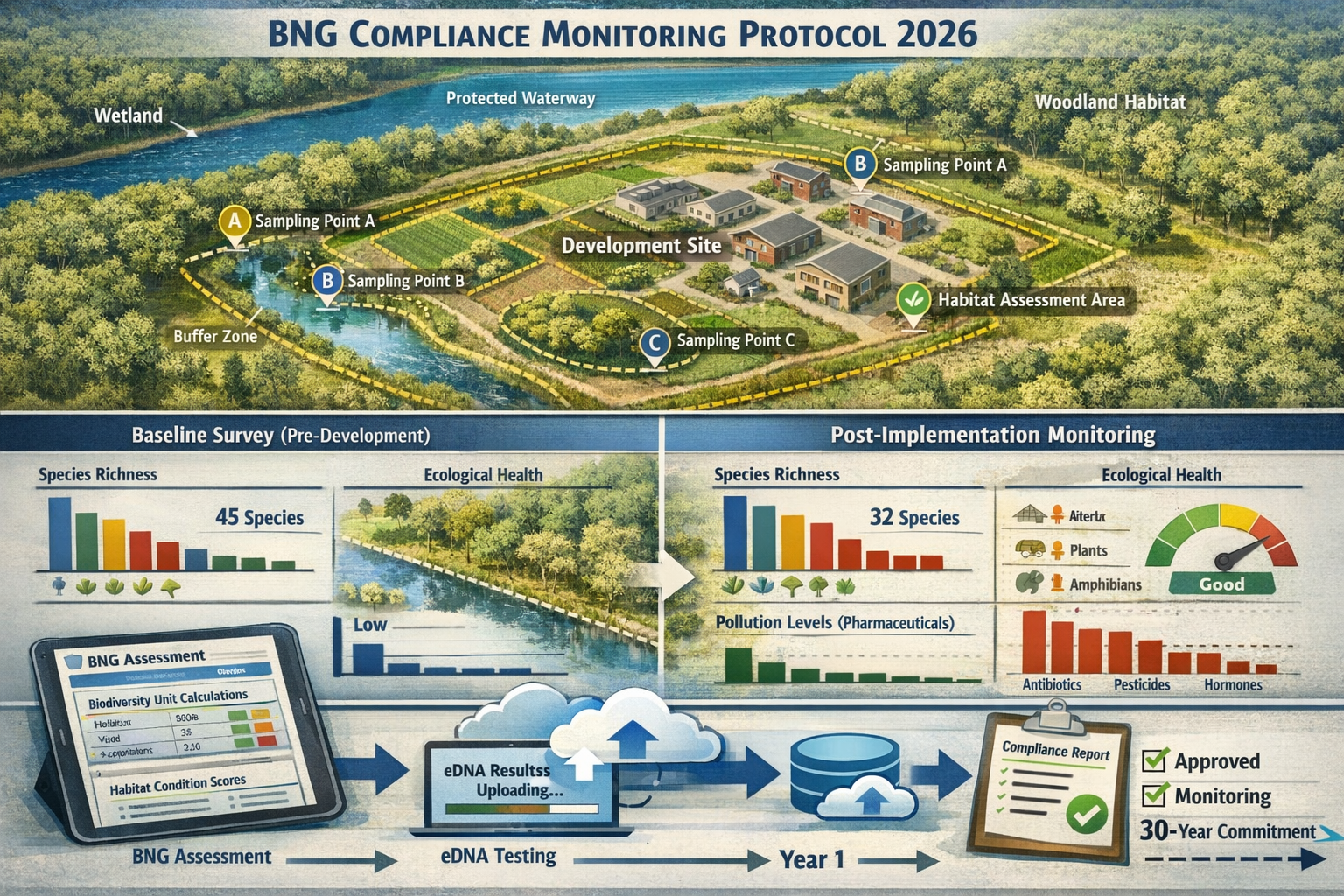
Best practice Pharmaceutical Pollutants in Biodiversity Surveys: eDNA Detection Strategies for Aquatic BNG Baselines employ an integrated monitoring framework that combines multiple detection approaches [2]:
Phase 1: Broad Survey (Metabarcoding)
- Collect water samples from representative habitat types across the site
- Process samples using universal primers targeting multiple taxonomic groups
- Generate comprehensive species inventories including fish, amphibians, invertebrates, and plants
- Identify bioindicator species sensitive to pharmaceutical contamination
Phase 2: Targeted Confirmation (Species-Specific qPCR)
- Deploy species-specific assays for protected or indicator species detected in Phase 1
- Conduct pharmaceutical metabolite screening for priority contaminants
- Quantify DNA concentration to estimate relative abundance
- Validate presence of sensitive species indicating low contamination
Phase 3: Verification and Validation
- Deploy camera traps or conduct visual surveys to confirm key species presence
- Collect tissue samples from captured specimens for pharmaceutical bioaccumulation analysis
- Document habitat condition using standardized assessment protocols
- Integrate findings into BNG metric calculations
Phase 4: Post-Implementation Monitoring
- Repeat eDNA sampling post-development to verify mitigation effectiveness
- Track pharmaceutical contamination levels over time
- Assess whether sensitive species persist or recolonize
- Document compliance with 30-year monitoring commitments
This closed-loop monitoring approach ensures that baseline data accurately reflects both biological diversity and chemical stressor presence—essential for achieving biodiversity net gain without risk.

Site Selection and Sampling Strategy
Strategic sampling design maximizes the value of eDNA surveys for pharmaceutical pollution detection. Priority sampling locations include:
| Location Type | Rationale | Sampling Frequency |
|---|---|---|
| Upstream reference sites | Establish background contamination levels | Single baseline + annual |
| Wastewater discharge points | Direct source identification | Quarterly monitoring |
| Site water features | On-site habitat baseline assessment | Pre/during/post development |
| Downstream monitoring stations | Track off-site contamination impacts | Quarterly during construction |
| Tributary confluences | Identify contamination pathways | Seasonal assessment |
Sampling timing should account for seasonal variation in pharmaceutical use patterns, flow regimes, and species activity periods. Winter sampling often reveals elevated pharmaceutical concentrations due to increased medication use and reduced dilution flows, while spring/summer sampling captures peak biodiversity during breeding seasons.
Laboratory Analysis and Interpretation
Selecting appropriate laboratory partners ensures data quality and regulatory acceptance. When commissioning eDNA analysis for BNG compliance, ecology surveyors should verify:
- Accreditation status – ISO 17025 or equivalent quality management certification
- Reference database coverage – Comprehensive UK species libraries for accurate identification
- Contamination controls – Rigorous protocols preventing false positives
- Quantification capabilities – qPCR expertise for pharmaceutical metabolite detection
- Reporting standards – Clear presentation of detection limits, confidence intervals, and species lists
Data interpretation requires ecological expertise to distinguish natural community variation from pollution-driven changes. eDNA surveys have identified a "protected area effect" with higher sensitive/threatened species occurrence inside protected areas [4]—providing data-driven validation that eDNA monitoring can detect real conservation outcomes. Similar analytical approaches can reveal pharmaceutical pollution signatures through reduced sensitive species detection and elevated pollution-tolerant generalist presence.
Integration with BNG Metric Calculations
Pharmaceutical contamination findings must inform habitat condition assessments within the BNG metric framework. Key considerations include:
Condition Assessment Adjustments:
- Presence of pharmaceutical-sensitive indicator species supports "good" condition scores
- Detection of contamination biomarkers justifies condition score reductions
- Absence of expected sensitive species despite suitable habitat suggests degraded conditions
- Elevated pollution-tolerant generalist dominance indicates functional impairment
Habitat Distinctiveness Considerations:
- Contaminated watercourses may not qualify for "high distinctiveness" classifications
- Pharmaceutical pollution presence limits habitat functionality regardless of physical structure
- Remediation requirements may affect on-site versus off-site delivery decisions
Risk Assessment and Mitigation:
- Pharmaceutical contamination increases ecological risk ratings
- Enhanced monitoring requirements may apply to contaminated baseline sites
- Remediation measures may be necessary before habitat creation/enhancement credit claims
For developers navigating these complexities, consulting with specialists experienced in creating biodiversity plans that address pharmaceutical pollution ensures compliance and reduces approval delays.
Regulatory Context and Future Directions
2026 Compliance Requirements
As of 2026, regulatory expectations for aquatic BNG baselines increasingly incorporate pharmaceutical pollution screening, particularly for development sites near:
- Wastewater treatment plant discharge zones
- Hospital or healthcare facility effluent outfalls
- Agricultural areas with intensive livestock operations
- Urban catchments with combined sewer overflow points
- Protected waterways designated for conservation priority species
While pharmaceutical screening is not yet universally mandated, proactive developers recognize that comprehensive baseline data prevents future compliance challenges. Local planning authorities increasingly question BNG assessments that fail to address known contamination risks, particularly where pharmaceutical pollution could undermine proposed habitat creation or enhancement measures.
Best Practice Recommendations for Ecology Surveyors
Professional ecology surveyors incorporating pharmaceutical pollution assessment into 2026 BNG surveys should adopt these protocols:
🔬 Screening Protocols:
- Include pharmaceutical metabolite analysis in aquatic eDNA sampling programs
- Target priority contaminants based on local pollution sources (wastewater, agriculture, industry)
- Establish reference site comparisons to quantify contamination gradients
- Document bioindicator species presence/absence as contamination proxies
📋 Reporting Standards:
- Explicitly state whether pharmaceutical screening was conducted
- Report detection limits and analytical methods for transparency
- Interpret biodiversity findings in context of contamination status
- Recommend monitoring frequencies based on pollution risk assessment
⚠️ Risk Communication:
- Clearly communicate pharmaceutical pollution risks to clients and stakeholders
- Explain implications for habitat functionality and BNG metric calculations
- Outline remediation options where contamination compromises baseline quality
- Document uncertainty where pharmaceutical screening was not conducted
🔄 Adaptive Management:
- Design monitoring programs that track contamination trends over time
- Establish trigger thresholds for management intervention
- Plan contingency measures if contamination increases post-development
- Integrate pharmaceutical screening into long-term BNG monitoring commitments
Emerging Technologies and Research Directions
The field of eDNA-based pharmaceutical pollution monitoring continues to evolve rapidly. Emerging developments include:
Advanced Detection Methods:
- Droplet digital PCR for ultra-sensitive pharmaceutical metabolite quantification
- Portable field sequencing devices enabling real-time on-site analysis
- Artificial intelligence algorithms for automated bioindicator community interpretation
- Multi-marker approaches combining species detection with stress biomarker identification
Expanded Monitoring Capabilities:
- Sediment eDNA analysis revealing historical contamination patterns
- Invertebrate eDNA targeting pharmaceutical-sensitive taxa
- Microbial community profiling indicating antibiotic resistance gene prevalence
- Cross-ecosystem monitoring tracking pharmaceutical transfer to terrestrial habitats
These technological advances will further enhance the value of Pharmaceutical Pollutants in Biodiversity Surveys: eDNA Detection Strategies for Aquatic BNG Baselines for comprehensive environmental assessment.
Practical Implementation Case Studies
Urban Development Near Wastewater Outfall
A residential development proposed adjacent to a river receiving treated wastewater discharge implemented comprehensive eDNA baseline surveys incorporating pharmaceutical screening. Initial metabarcoding detected 47 aquatic species but revealed concerning absence of pharmaceutical-sensitive mayfly families expected in the habitat type.
Targeted qPCR analysis confirmed elevated estrogen and antidepressant metabolite concentrations. The developer adjusted the BNG strategy to:
- Downgrade baseline habitat condition scores reflecting functional impairment
- Increase biodiversity unit requirements to account for degraded starting conditions
- Implement enhanced buffer zones with pharmaceutical-filtering wetland features
- Establish quarterly pharmaceutical monitoring throughout the 30-year commitment period
This proactive approach prevented future compliance challenges and demonstrated environmental responsibility to planning authorities, facilitating approval.
Agricultural Catchment Restoration Project
A landowner seeking to establish a habitat bank in an agricultural catchment used eDNA surveys to document baseline conditions. Pharmaceutical screening revealed veterinary antibiotic contamination from upstream livestock operations, with associated shifts toward pollution-tolerant fish communities.
The eDNA data informed a two-phase restoration strategy:
- Source control – Collaboration with upstream farmers to implement improved manure management
- Habitat enhancement – Creation of riparian buffer wetlands providing natural pharmaceutical filtration
Follow-up eDNA monitoring demonstrated pharmaceutical concentration reductions and recolonization by sensitive species, supporting higher habitat condition scores and increased biodiversity unit values for the habitat bank.
Linear Infrastructure Baseline Assessment
A highway expansion project crossing multiple watercourses deployed rapid eDNA surveys to establish baselines across 15 stream crossings. Metabarcoding provided comprehensive species inventories within tight project timelines, while pharmaceutical screening identified two contamination hotspots requiring enhanced mitigation.
The integrated approach enabled:
- Efficient baseline documentation meeting BNG requirements
- Targeted mitigation design addressing site-specific contamination risks
- Defensible condition assessments incorporating chemical stressor data
- Cost-effective monitoring covering extensive linear infrastructure
This case demonstrates the scalability advantages of eDNA approaches for large, complex development projects requiring comprehensive biodiversity impact assessments.
Conclusion
Pharmaceutical Pollutants in Biodiversity Surveys: eDNA Detection Strategies for Aquatic BNG Baselines represent an essential evolution in environmental assessment practice for 2026 and beyond. The convergence of widespread pharmaceutical contamination, mandatory BNG requirements, and advanced eDNA detection technologies creates both challenges and opportunities for developers, landowners, and ecology professionals.
Traditional biodiversity surveys that ignore pharmaceutical pollution risk significantly overestimating habitat quality and ecological functionality. Hormone disruptors, antibiotics, and psychoactive compounds documented across English waterways fundamentally alter aquatic ecosystems—feminizing fish, modifying behavior, disrupting food webs, and transferring contamination to terrestrial predators through insect emergence. These invisible stressors undermine biodiversity value regardless of species lists or physical habitat structure.
eDNA metabarcoding offers a powerful solution, enabling simultaneous species detection and pharmaceutical impact assessment through integrated monitoring frameworks. The technology's cost-effectiveness, scalability, and multi-functional biomonitoring capabilities make it ideally suited for comprehensive BNG baseline establishment. By combining broad metabarcoding surveys with targeted pharmaceutical metabolite screening, ecology surveyors can provide clients with accurate, defensible baseline data that withstands regulatory scrutiny and supports long-term project success.
Actionable Next Steps
For Developers:
- Require pharmaceutical screening in aquatic eDNA baseline surveys for sites near contamination sources
- Budget for integrated monitoring approaches combining species detection and chemical analysis
- Engage ecology consultants experienced with pharmaceutical pollution assessment
- Consider contamination risks when evaluating on-site versus off-site BNG delivery options
For Landowners:
- Establish pharmaceutical contamination status before marketing habitat banking opportunities
- Invest in comprehensive eDNA baseline surveys to maximize biodiversity unit values
- Address contamination sources to enhance habitat quality and marketability
- Maintain pharmaceutical monitoring data demonstrating habitat integrity over time
For Ecology Professionals:
- Develop competency in eDNA survey design and pharmaceutical pollution interpretation
- Establish relationships with accredited laboratories offering integrated analysis services
- Incorporate pharmaceutical screening protocols into standard aquatic survey methodologies
- Educate clients about contamination risks and monitoring value propositions
For Planning Authorities:
- Request pharmaceutical contamination data for aquatic BNG assessments near pollution sources
- Evaluate whether baseline habitat condition assessments adequately account for chemical stressors
- Require long-term pharmaceutical monitoring where contamination risks threaten BNG outcomes
- Support proactive developers implementing comprehensive baseline characterization
The integration of pharmaceutical pollution assessment into biodiversity surveying reflects the maturation of environmental practice toward holistic ecosystem evaluation. As regulatory expectations evolve and detection technologies advance, early adopters of these integrated approaches will benefit from streamlined approvals, reduced compliance risks, and genuine environmental outcomes that support both development objectives and biodiversity conservation.
For expert guidance on implementing Pharmaceutical Pollutants in Biodiversity Surveys: eDNA Detection Strategies for Aquatic BNG Baselines in your projects, contact experienced biodiversity surveyors who can design monitoring programs tailored to your site-specific requirements and regulatory context.
References
[1] Insights in Pharmaceutical Pollution: The Prospective Role of eDNA – https://pubmed.ncbi.nlm.nih.gov/37999555/
[2] eDNA Testing for Invasive Species (2026): What Works, What Doesn't – https://landwildlifereport.com/2026/01/20/edna-testing-for-invasive-species/
[3] New roadmap outlines strategies to reduce pharmaceutical pollution – https://phys.org/news/2026-01-roadmap-outlines-strategies-pharmaceutical-pollution.html
[4] Closing the Gap Between Biodiversity Commitments and Measuring Nature – https://sps.columbia.edu/news/closing-gap-between-biodiversity-commitments-and-measuring-nature
[5] Environmental Science & Technology – https://pubs.acs.org/doi/10.1021/acs.est.5c09855
[6] Insects are moving pharmaceutical pollutants from rivers to land – https://news.mongabay.com/2026/02/insects-are-moving-pharmaceutical-pollutants-from-rivers-to-land-risks-unknown/
[7] Environmental DNA Services – https://www.sgs.com/en-us/services/e-dna


