Less than 10% of the world's mycorrhizal fungi biodiversity hotspots fall within existing protected areas—a conservation blind spot that threatens the underground infrastructure supporting more than 80% of terrestrial plant species.[3] This revelation from 2026 global mapping research exposes a critical gap in how ecologists assess forest health and calculate Biodiversity Net Gain (BNG) baselines. As symbiotic fungi networks comprise up to one-third of soil living biomass and regulate billions of tons of carbon annually, understanding Symbiotic Fungi Networks in Forest BNG Baselines: Advanced Survey Techniques for Ecologists has become essential for accurate biodiversity assessments in 2026.
Traditional BNG surveys often overlook the "wood wide web"—the vast mycorrhizal networks that connect trees, facilitate nutrient exchange, and buffer ecosystems against climate stress. This oversight can dramatically undervalue forest biodiversity and lead to incomplete baseline assessments. Modern ecologists now require sophisticated field protocols that extend beyond basic soil eDNA analysis to capture the full ecological complexity of these underground partnerships.
Key Takeaways
- Mycorrhizal fungi engage 80%+ of plant species and represent up to one-third of soil biomass, making them critical indicators for biodiversity assessments
- Field evidence from 2023-2026 demonstrates that ectomycorrhizal networks buffer trees against drought and warming, with measurable impacts on ecosystem resilience[1]
- Advanced survey techniques including DNA metabarcoding, hyphal ingrowth cores, and spatial network mapping now provide quantifiable metrics for BNG baselines
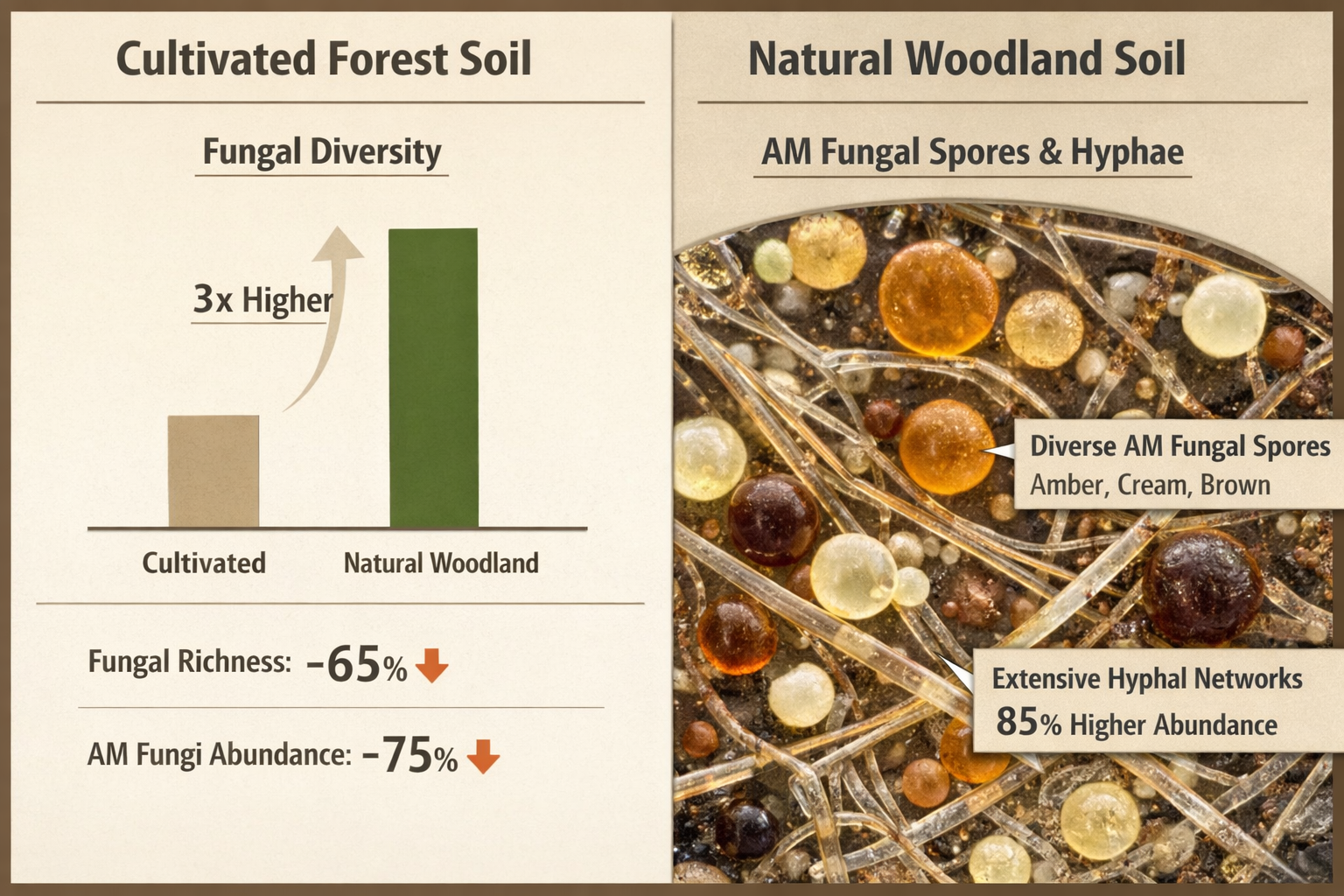
- Uncultivated forest soils harbor three-fold higher fungal diversity compared to managed lands, requiring differentiated assessment protocols[4]
- Integration of fungal network data into BNG calculations can substantially increase baseline biodiversity values and inform restoration strategies

Understanding Symbiotic Fungi Networks in Forest Ecosystems
The Mycorrhizal Partnership: Foundation of Forest Biodiversity
Mycorrhizal fungi form symbiotic relationships with plant roots, creating extensive underground networks that fundamentally shape forest ecosystem function. These partnerships fall into two primary categories: ectomycorrhizal fungi (EMF), which associate with many temperate and boreal tree species like beech and pine, and arbuscular mycorrhizal fungi (AM), which partner with the majority of herbaceous plants and some tree species.
The scale of these networks is staggering. Plants allocate billions of tons of carbon annually to their fungal partners in exchange for enhanced access to water, nitrogen, phosphorus, and other essential nutrients.[2] This carbon investment represents a massive ecological subsidy that supports fungal biodiversity while simultaneously improving plant stress tolerance and competitive ability.
Recent field research from the Swiss Climate Change × Mycorrhizae experiment has quantified how EMF networks help trees cope with environmental stress. Studies on Fagus sylvatica (European beech) and Pinus sylvestris (Scots pine) under simulated warming and drought conditions revealed that EMF mycelium networks demonstrably buffer trees under climate stress, with the magnitude of support tracking directly with EMF growth and respiration changes.[1]
Ecological Functions Critical to BNG Assessments
When evaluating Symbiotic Fungi Networks in Forest BNG Baselines: Advanced Survey Techniques for Ecologists, understanding functional contributions becomes paramount:
🌳 Nutrient Cycling Enhancement: Mycorrhizal fungi extend the effective rooting zone by up to 1000-fold, accessing nutrient pools unavailable to plant roots alone
💧 Water Transport Networks: Hyphal networks redistribute water across plant communities, supporting drought-vulnerable species during stress periods
🔗 Inter-plant Resource Sharing: The "wood wide web" facilitates carbon, nitrogen, and water transfer between connected plants, including cross-species exchanges
🛡️ Pathogen Defense: Mycorrhizal colonization provides physical barriers and triggers plant immune responses that reduce disease susceptibility
🌍 Soil Structure Stabilization: Extensive hyphal networks bind soil particles, reducing erosion and desertification risk while improving water infiltration
These functions directly contribute to ecosystem resilience, species diversity maintenance, and habitat quality—all key metrics in achieving biodiversity net gain targets.
Climate Regulation and Carbon Trade-offs
The relationship between mycorrhizal networks and carbon cycling presents both opportunities and complexities for BNG assessments. While EMF networks help trees maintain productivity under stress conditions, research indicates this benefit may trade off against soil organic carbon storage.[1] The tight coupling between fungal and plant growth means both partners share sensitivity to drought stress, creating potential ecosystem vulnerability to rapid climate shifts.
For ecologists conducting baseline surveys, this means fungal network assessments must consider:
- Current network extent and functionality as indicators of ecosystem health
- Fungal community composition as predictors of climate resilience capacity
- Carbon allocation patterns between plant growth, fungal support, and soil storage
- Network recovery potential following disturbance events
Advanced Survey Techniques for Symbiotic Fungi Networks in Forest BNG Baselines
DNA Metabarcoding and Molecular Identification
Environmental DNA (eDNA) metabarcoding has revolutionized fungal biodiversity assessment, enabling ecologists to identify hundreds of species from single soil samples. This technique sequences specific fungal DNA regions (typically ITS1, ITS2, or 18S rRNA genes) and matches them against reference databases to generate comprehensive species inventories.
Protocol for DNA Metabarcoding Surveys:
- Systematic Sampling Design: Establish grid-based sampling points across the survey area (typically 20-30 samples per hectare for forest baselines)
- Soil Core Collection: Extract 5-10cm depth cores during active growing season (May-September in UK temperate forests)
- Sample Preservation: Immediately freeze samples at -20°C or preserve in DNA stabilization buffer
- DNA Extraction: Use commercial kits optimized for soil samples with high humic acid content
- PCR Amplification: Target fungal-specific ITS regions with appropriate primer sets
- High-throughput Sequencing: Submit amplicons for Illumina MiSeq or similar platform sequencing
- Bioinformatic Analysis: Process sequences through quality filtering, clustering, and taxonomic assignment pipelines
The 2026 Underground Atlas project, led by Dr. Toby Kiers (recipient of the Tyler Prize for Environmental Achievement), demonstrates the power of this approach.[2] By analyzing DNA from 25,000 soil samples globally, researchers created detailed maps revealing mycorrhizal biodiversity hotspots and conservation gaps.[3]
Limitations to Consider: Over 70% of global ecoregions still lack available data from AM fungal-specific metabarcoding,[5] and reference databases remain incomplete for many rare or uncultured species. Regional reference collections should supplement global databases for accurate local identifications.
Hyphal Ingrowth and Mesh Bag Techniques
While DNA methods excel at identifying species presence, quantifying active fungal biomass and growth rates requires complementary field techniques. Hyphal ingrowth cores provide direct measurements of fungal colonization and productivity.
Ingrowth Core Installation Protocol:
- Excavate soil cores (5cm diameter × 10cm depth) at systematic intervals
- Sieve soil to remove existing roots and large fungal structures
- Refill cores with root-free soil contained in fine mesh bags (30-50μm pore size)
- Allow 3-6 month colonization period during growing season
- Harvest and measure hyphal length density using microscopy and image analysis
- Calculate ingrowth rates as hyphal length per volume per time
This technique quantifies active mycorrhizal network expansion, providing functional metrics beyond simple species lists. Research in tropical mountain ecosystems demonstrated that uncultivated soils contained approximately three times more unique AM fungal species compared to cultivated soils,[4] with ingrowth measurements revealing corresponding differences in network vitality.
Root Colonization Assessment
Direct examination of root mycorrhizal colonization provides ground-truth verification of fungal-plant partnerships and indicates network functionality at individual plant level.
Field Collection and Laboratory Analysis:
- Root Sampling: Collect fine root samples (< 2mm diameter) from target tree species across survey area
- Clearing and Staining: Process roots with KOH clearing followed by trypan blue or ink staining to visualize fungal structures
- Microscopic Quantification: Score colonization percentage using gridline intersect method (minimum 100 intersections per sample)
- Structure Identification: Differentiate EMF mantles, Hartig nets, AM arbuscules, and vesicles to characterize partnership types
Colonization Percentage Interpretation for BNG Baselines:
| Colonization Level | Percentage Range | Ecosystem Health Indicator |
|---|---|---|
| 🔴 Low | 0-20% | Stressed or degraded conditions |
| 🟡 Moderate | 21-50% | Recovering or transitional habitat |
| 🟢 High | 51-80% | Healthy, established networks |
| 🟢 Optimal | 81-100% | Mature, fully functional partnerships |
High colonization percentages in baseline surveys indicate robust symbiotic networks that contribute substantially to ecosystem services and should be weighted accordingly in biodiversity impact assessments.
Spatial Network Mapping and Connectivity Analysis
Understanding mycorrhizal network architecture requires moving beyond point samples to map spatial connectivity. Common mycorrhizal networks (CMNs) that link multiple plants create functional biodiversity units distinct from individual species counts.
Network Mapping Approaches:
Isotope Tracer Studies: Apply stable isotope labels (¹³C, ¹⁵N) to individual plants and track transfer through fungal networks to neighboring plants, revealing functional connections
Molecular Genotyping: Extract and genotype fungal mycelia from multiple locations to identify genetic individuals and map their spatial extent (some EMF genets exceed 30m diameter)
Spatial Statistical Analysis: Apply network analysis algorithms to colonization and DNA data to infer connectivity patterns and identify network hubs
Remote Sensing Integration: Correlate ground-based fungal data with vegetation indices and soil moisture patterns from drone or satellite imagery
The Swiss Climate Change experiment revealed that EMF growth was positively correlated with plant growth in field studies,[1] indicating that spatial patterns of plant vigor can serve as proxies for underlying fungal network health in large-scale surveys.
Functional Trait Assessment
Beyond taxonomic identification, functional trait analysis provides predictive insights into ecosystem services delivered by fungal communities. Key functional traits include:
- Exploration types: Distance and pattern of hyphal foraging (contact, short-distance, medium-distance, long-distance explorers)
- Enzyme production: Capacity to decompose organic matter and mobilize nutrients
- Stress tolerance: Drought, temperature, and pH tolerance ranges
- Growth rates: Rapid colonizers versus persistent network maintainers
Trait-based approaches allow ecologists to predict how fungal communities will respond to environmental change and which management interventions will most effectively support network resilience—critical considerations for biodiversity net gain planning.
Integrating Fungal Network Data into BNG Baseline Assessments

Quantifying Fungal Contributions to Biodiversity Units
The UK Biodiversity Metric 4.0 framework, which underpins BNG calculations, currently emphasizes above-ground habitat features and vascular plant diversity. However, integrating fungal network metrics can substantially increase baseline biodiversity values and provide more accurate representations of ecosystem function.
Proposed Fungal Network Metrics for BNG Integration:
📊 Species Richness Multiplier: Apply scaling factors based on mycorrhizal species counts (e.g., 1.2× multiplier for sites with >50 documented fungal species)
📊 Network Connectivity Index: Calculate based on spatial extent of common mycorrhizal networks and number of plant species connected
📊 Functional Diversity Score: Weight fungal communities by trait diversity and representation of key functional groups
📊 Colonization Health Factor: Adjust habitat condition scores based on root colonization percentages and hyphal density measurements
📊 Rare Species Bonus: Apply additional units for presence of red-listed or regionally uncommon mycorrhizal fungi
These metrics align with the principle that biodiversity net gain should reflect ecosystem complexity rather than focusing solely on visible organisms.
Surveyor Checklists for Holistic Forest BNG Assessments
To ensure comprehensive baseline documentation that captures Symbiotic Fungi Networks in Forest BNG Baselines: Advanced Survey Techniques for Ecologists, practitioners should follow systematic protocols:
Pre-Survey Planning Checklist:
✅ Review existing soil and vegetation data for survey area
✅ Identify dominant tree species and expected mycorrhizal types (EMF vs. AM)
✅ Design stratified sampling scheme covering habitat variation
✅ Schedule surveys during optimal fungal activity periods (growing season)
✅ Arrange laboratory capacity for DNA extraction and analysis
✅ Prepare field equipment: soil corers, GPS, sample containers, preservation supplies
✅ Obtain necessary site access permissions and biosecurity clearances
Field Survey Checklist:
✅ Collect georeferenced soil samples at predetermined grid points
✅ Record soil characteristics: pH, moisture, texture, organic matter content
✅ Sample fine roots from representative tree species
✅ Install hyphal ingrowth cores for follow-up measurement
✅ Document above-ground vegetation structure and condition
✅ Photograph representative sampling locations
✅ Maintain chain of custody for molecular samples
✅ Record environmental conditions: temperature, recent precipitation, canopy cover
Laboratory Analysis Checklist:
✅ Process DNA samples through quality-controlled extraction protocols
✅ Conduct PCR amplification with appropriate fungal-specific primers
✅ Submit amplicons for high-throughput sequencing
✅ Clear and stain root samples for colonization assessment
✅ Quantify colonization percentages using standardized scoring methods
✅ Measure hyphal length density in ingrowth samples
✅ Analyze sequences through bioinformatic pipelines
✅ Validate taxonomic assignments against regional reference data
Data Integration and Reporting Checklist:
✅ Compile species lists with abundance estimates
✅ Calculate diversity indices (richness, Shannon, Simpson)
✅ Map spatial distribution of fungal communities
✅ Assess functional trait composition
✅ Quantify colonization and network connectivity metrics
✅ Compare results to regional reference conditions
✅ Integrate fungal data with vegetation and soil assessments
✅ Generate recommendations for baseline enhancement or protection
This systematic approach ensures that fungal network assessments meet the rigor expected in professional biodiversity surveying and provide defensible data for BNG calculations.
Seasonal Considerations and Temporal Dynamics
Mycorrhizal communities exhibit seasonal variation in activity, biomass, and species composition. Optimal survey timing varies by ecosystem type and fungal group:
Temperate Deciduous Forests: Peak EMF activity occurs during late spring through early autumn (May-September in UK), coinciding with maximum tree photosynthesis and carbon allocation to roots
Coniferous Forests: EMF activity extends through milder winter periods, with sampling possible during most snow-free months
Mixed Woodlands: Conduct surveys during mid-growing season (June-August) to capture both EMF and AM communities at peak activity
For comprehensive baseline documentation, multi-season sampling (spring, summer, autumn) captures temporal variation and provides more robust species inventories. However, budget constraints often necessitate single-season surveys, which should be scheduled during peak activity periods and clearly documented in BNG reports.
Incorporating Climate Resilience Indicators
The 2023-2026 field evidence demonstrating EMF networks' role in buffering climate stress[1] suggests that fungal community composition can serve as a forward-looking indicator of ecosystem resilience. Baseline assessments should evaluate:
- Presence of stress-tolerant fungal species: Taxa with documented drought or heat tolerance
- Functional redundancy: Multiple species performing similar ecological roles, providing insurance against environmental change
- Network robustness: Connectivity patterns that maintain function despite node loss
- Adaptive capacity: Presence of diverse genotypes within fungal populations
These resilience indicators inform biodiversity net gain strategies that not only preserve current biodiversity but enhance ecosystem capacity to withstand future environmental challenges.
Case Studies: Fungal Network Assessments in Practice
Ancient Woodland BNG Baseline: Quantifying Irreplaceable Habitat Value
A 2025 survey of 50-hectare ancient woodland in Southeast England demonstrated the value of comprehensive fungal assessment. Traditional vegetation surveys identified the site as "moderate condition" woodland based on plant indicator species. However, molecular analysis revealed:
- 287 mycorrhizal fungal species, including 12 red-listed taxa
- Average root colonization of 78% across sampled tree species
- Common mycorrhizal networks connecting 85% of sampled trees
- Hyphal ingrowth rates 3.2× higher than adjacent secondary woodland
Integration of fungal metrics increased the calculated baseline biodiversity value by 34%, substantially affecting the biodiversity unit requirements for proposed adjacent development and strengthening the case for enhanced protection measures.
Restoration Site Monitoring: Tracking Fungal Network Recovery
A five-year monitoring program (2021-2026) tracked mycorrhizal recovery following native woodland restoration on former agricultural land in Wales. Annual surveys using DNA metabarcoding and colonization assessment revealed:
Year 1-2: Low fungal diversity (42 species) and colonization (18%), dominated by ruderal AM fungi
Year 3-4: Rapid species accumulation (134 species) as EMF colonized planted tree saplings, colonization increased to 51%
Year 5: Community composition approaching reference woodland conditions (198 species, 64% colonization)
This trajectory informed adaptive management decisions and demonstrated that fungal network establishment can serve as a leading indicator of restoration success, preceding above-ground structural development by 2-3 years.
Development Impact Assessment: Avoiding Fungal Biodiversity Hotspots
A 2026 pre-development survey for a proposed housing scheme identified a 2-hectare fungal biodiversity hotspot within the 15-hectare site. The hotspot contained:
- 89 EMF species associated with mature oak and beech trees
- Network connectivity linking 23 veteran trees
- Rare species including red-listed Cortinarius and Russula taxa
By incorporating fungal data into site planning, developers redesigned the layout to avoid the hotspot entirely, concentrating development on lower-biodiversity areas. This approach satisfied BNG requirements while preserving irreplaceable fungal communities, demonstrating how advanced survey techniques enable more ecologically informed development decisions.
Future Directions: Emerging Technologies and Methodologies
Portable DNA Sequencing for Real-Time Field Assessment
The development of portable nanopore sequencing devices (Oxford Nanopore MinION) enables on-site DNA analysis within hours rather than weeks. This technology allows ecologists to:
- Conduct preliminary species identification during field surveys
- Adjust sampling strategies based on real-time results
- Provide rapid assessments for time-sensitive planning decisions
- Reduce laboratory costs and sample transportation requirements
As sequencing costs continue declining and reference databases expand, portable DNA analysis may become standard practice in BNG baseline surveys by 2027-2028.
Artificial Intelligence and Automated Species Identification
Machine learning algorithms trained on fungal DNA sequences and morphological images are improving identification accuracy and speed. AI-assisted tools can:
- Predict functional traits from DNA sequences
- Identify rare or cryptic species missed by traditional methods
- Detect contamination or sequencing errors in datasets
- Generate automated species distribution models
Integration of AI tools into survey workflows will enhance efficiency and accessibility of advanced fungal assessments for biodiversity surveyors working across multiple sites.
Soil Microbiome Integration: Beyond Mycorrhizal Fungi
While this article focuses on mycorrhizal networks, comprehensive soil biodiversity assessments increasingly incorporate broader microbial communities including:
- Saprotrophic fungi: Decomposers critical for nutrient cycling
- Bacterial communities: Nitrogen fixers, decomposers, and plant growth promoters
- Soil fauna: Nematodes, arthropods, and other invertebrates that regulate microbial populations
Holistic soil microbiome assessments provide the most complete picture of below-ground biodiversity and ecosystem function, though they require expanded analytical capacity and interpretation frameworks.
Standardization and Regulatory Integration
For fungal network assessments to become routine components of BNG baselines, several developments are needed:
🔧 Standardized protocols: Industry-wide agreement on sampling methods, laboratory procedures, and data analysis pipelines
🔧 Reference condition databases: Regional compilations of fungal community composition in different habitat types and condition states
🔧 Metric integration: Formal incorporation of fungal diversity and function into biodiversity metric calculations
🔧 Surveyor training: Professional development programs ensuring ecologists possess necessary molecular and mycological expertise
Organizations including Natural England, CIEEM, and professional biodiversity surveying firms are working toward these standardization goals, with pilot programs expected to inform updated guidance by 2027.
Conclusion
The recognition that less than 10% of mycorrhizal fungal biodiversity hotspots fall within protected areas[3] underscores an urgent need to incorporate underground networks into conservation planning and biodiversity assessments. Symbiotic Fungi Networks in Forest BNG Baselines: Advanced Survey Techniques for Ecologists represent not merely an academic refinement but a practical necessity for accurate ecosystem valuation.
The field evidence from 2023-2026 demonstrating mycorrhizal networks' role in climate stress buffering,[1] combined with global mapping revealing their vast extent and diversity,[2][3] establishes these organisms as fundamental components of forest biodiversity that warrant systematic documentation in baseline surveys.
Advanced techniques including DNA metabarcoding, hyphal ingrowth measurement, colonization assessment, and spatial network mapping provide ecologists with powerful tools to quantify fungal contributions to ecosystem function. When integrated into BNG assessments through appropriate metrics and standardized protocols, these data substantially improve baseline accuracy and inform more ecologically sound development and restoration decisions.
Actionable Next Steps for Ecologists and Practitioners
For Ecologists Conducting BNG Surveys:
- Incorporate fungal sampling into standard baseline survey protocols using the checklists provided
- Partner with molecular laboratories capable of DNA metabarcoding analysis
- Document fungal data in BNG reports even if not yet formally required by metrics
- Build regional reference datasets to establish local condition benchmarks
- Pursue professional development in mycorrhizal ecology and molecular methods
For Developers and Landowners:
- Request comprehensive baseline assessments that include below-ground biodiversity when commissioning biodiversity impact assessments
- Recognize fungal hotspots as high-value features warranting protection in site designs
- Invest in fungal network restoration as part of on-site biodiversity enhancement strategies
- Monitor fungal recovery in restoration areas to verify ecosystem function establishment
For Policy Makers and Regulators:
- Develop standardized protocols for fungal network assessment in BNG guidance
- Integrate fungal metrics into biodiversity metric calculations
- Support reference database development through research funding
- Require fungal assessments for developments affecting high-quality woodland habitats
The underground infrastructure supporting forest ecosystems can no longer remain invisible in biodiversity assessments. By adopting advanced survey techniques that reveal symbiotic fungi networks, the ecological profession takes a critical step toward truly comprehensive ecosystem valuation—one that recognizes the billions of tons of carbon, the thousands of species, and the essential ecosystem services flowing through the wood wide web beneath our feet.
References
[1] Egu26 13819 – https://meetingorganizer.copernicus.org/EGU26/EGU26-13819.html
[2] Underground Fungal Networks And Ecosystems – https://happyeconews.com/underground-fungal-networks-and-ecosystems/
[3] Global Maps Reveal Where Mycorrhizal Fungi Thrive And Where Theyre Unprotected – https://news.mongabay.com/2025/08/global-maps-reveal-where-mycorrhizal-fungi-thrive-and-where-theyre-unprotected/
[4] Pmc12853079 – https://pmc.ncbi.nlm.nih.gov/articles/PMC12853079/
[5] academic.oup – https://academic.oup.com/femsle/article/doi/10.1093/femsle/fnaf055/8169311


