The way we measure and understand biodiversity is undergoing a radical transformation. Traditional survey methods—visual counts, camera traps, and physical specimen collection—are giving way to revolutionary technologies that can detect species without ever seeing them. The Radical Transformation in Biodiversity Measurement: eDNA and Acoustic Protocols for 2026 Ecology Surveys represents a paradigm shift that promises to revolutionize how developers, ecologists, and land managers approach biodiversity monitoring and achieve meaningful conservation outcomes. 🌍
In 2026, as Biodiversity Net Gain (BNG) requirements become increasingly stringent across England, the need for accurate, comprehensive, and efficient biodiversity assessment has never been greater. Environmental DNA (eDNA) analysis and bioacoustic monitoring offer unprecedented capabilities to detect elusive species, track temporal patterns, and establish robust environmental baselines—all while reducing survey disturbance and operational costs.
Key Takeaways
- eDNA technology can detect rare, endangered, and invasive species from a single water, soil, or air sample, capturing genetic traces that traditional surveys miss entirely
- Bioacoustic monitoring provides continuous, non-invasive data collection that tracks seasonal patterns, habitat use, and behavioral changes across large geographic areas
- Combined protocols strengthen environmental baselines and enable faster, more reliable change detection to support biodiversity net gain strategies and compliance
- Automated sampling technologies like the DOT-NM Autosampler eliminate human interaction requirements, significantly enhancing safety and scalability in field operations
- Global standardization efforts through initiatives like eDNA Expeditions 2026-2028 are establishing reproducible workflows and open-access data sharing across 25 marine sites worldwide
Understanding the Radical Transformation in Biodiversity Measurement
What Makes This Transformation Radical?
The shift toward molecular and acoustic biodiversity measurement represents more than incremental improvement—it fundamentally changes what ecologists can detect, when they can detect it, and how they interpret ecosystem health. Traditional visual surveys require skilled observers to physically encounter organisms, creating inherent limitations in detecting nocturnal, cryptic, or rare species. These methods also introduce observer bias, weather dependencies, and significant time investments.
Environmental DNA (eDNA) changes this equation entirely. Every organism constantly sheds genetic material through skin cells, scales, mucus, feces, and other biological traces. A single water sample from a pond can reveal the presence of dozens of fish, amphibian, and invertebrate species without capturing a single individual.[3] Soil samples can detect fungi, bacteria, plants, and soil-dwelling animals. Even air samples can capture pollen, spores, and environmental traces from birds and mammals.
Bioacoustic monitoring complements eDNA by answering different questions. While eDNA tells us "who is here," acoustics reveals "when are they active," "what behaviors are occurring," and "how is the soundscape changing over time."[1] Automated recording units can operate continuously for months, capturing vocalizations from birds, bats, amphibians, marine mammals, and even fish—creating temporal datasets impossible to achieve through manual surveys.
The Science Behind eDNA Detection
The technical foundation of eDNA analysis involves several critical steps:
- Sample Collection: Water, soil, or air is filtered through specialized membranes (typically 0.22-0.45 μm for aquatic systems) that capture cellular material and free-floating DNA[3]
- DNA Extraction: Genetic material is isolated from the filter using standardized protocols like PCI/DNeasy methods
- Amplification: Target DNA sequences are multiplied using polymerase chain reaction (PCR) techniques
- Sequencing: Modern high-throughput sequencing identifies species by matching genetic sequences to reference databases
- Bioinformatic Analysis: Sophisticated algorithms process raw sequence data to generate species lists and relative abundance estimates
This "tree-of-life" bioinformatic workflow can detect biodiversity across all domains—from microscopic bacteria to megafauna—providing a comprehensive ecosystem snapshot from minimal physical sampling.[2]
eDNA and Acoustic Protocols Revolutionizing 2026 Ecology Surveys

Standardized Protocols for Reproducible Results
One of the most significant advances in the Radical Transformation in Biodiversity Measurement: eDNA and Acoustic Protocols for 2026 Ecology Surveys is the establishment of standardized methodologies that ensure data quality and comparability across different sites and survey teams.
Standardized eDNA filtration protocols now specify:
- Membrane specifications: 0.22-0.45 μm pore size for aquatic systems
- Sample volume: Typically 1-2 liters for freshwater, adjusted for marine environments
- Extraction methods: PCI/DNeasy for soils, MD8 samplers for atmospheric collection[3]
- Contamination controls: Rigorous field blanks and negative controls
- Storage conditions: Immediate preservation at -20°C or with stabilization buffers
These protocols address previous concerns about reproducibility and enable meaningful comparisons between baseline surveys and post-development monitoring—critical for demonstrating biodiversity net gain compliance.
Automated Sampling Technologies
The DOT-NM Autosampler, developed through collaboration between Dartmouth Ocean Technologies and NatureMetrics, represents a breakthrough in field deployment capabilities. This technology enables:
- Programmable collection schedules: Samples can be collected at specific times or intervals without human presence
- Self-cleaning mechanisms: Automated flushing prevents cross-contamination between samples
- Remote operation: Deployments in hazardous or difficult-to-access locations
- Enhanced safety: Eliminates need for repeated field visits in challenging environments[1]
For developers working on infrastructure projects near sensitive habitats, automated sampling dramatically reduces operational disruption while maintaining rigorous monitoring standards.
Bioacoustic Monitoring Systems
Modern acoustic monitoring has evolved far beyond simple recording devices. Integrated platforms like the SEAWATCH® Wind Lidar Buoy combine:
- Continuous soundscape monitoring: 24/7 recording captures all acoustic activity
- Multi-sensor integration: Simultaneous collection of genetic, video, and geophysical data
- Long-term deployment capability: Months of autonomous operation
- Real-time data transmission: Cloud-based processing and analysis[1]
Research demonstrates that acoustic monitoring alone can achieve approximately 52 species diversity detection after roughly 35 sampling days, establishing a strong baseline for comparative analysis.[4] When combined with eDNA protocols, detection capabilities increase substantially while providing complementary temporal and spatial information.
Implementing Combined eDNA and Acoustic Approaches for Maximum Impact
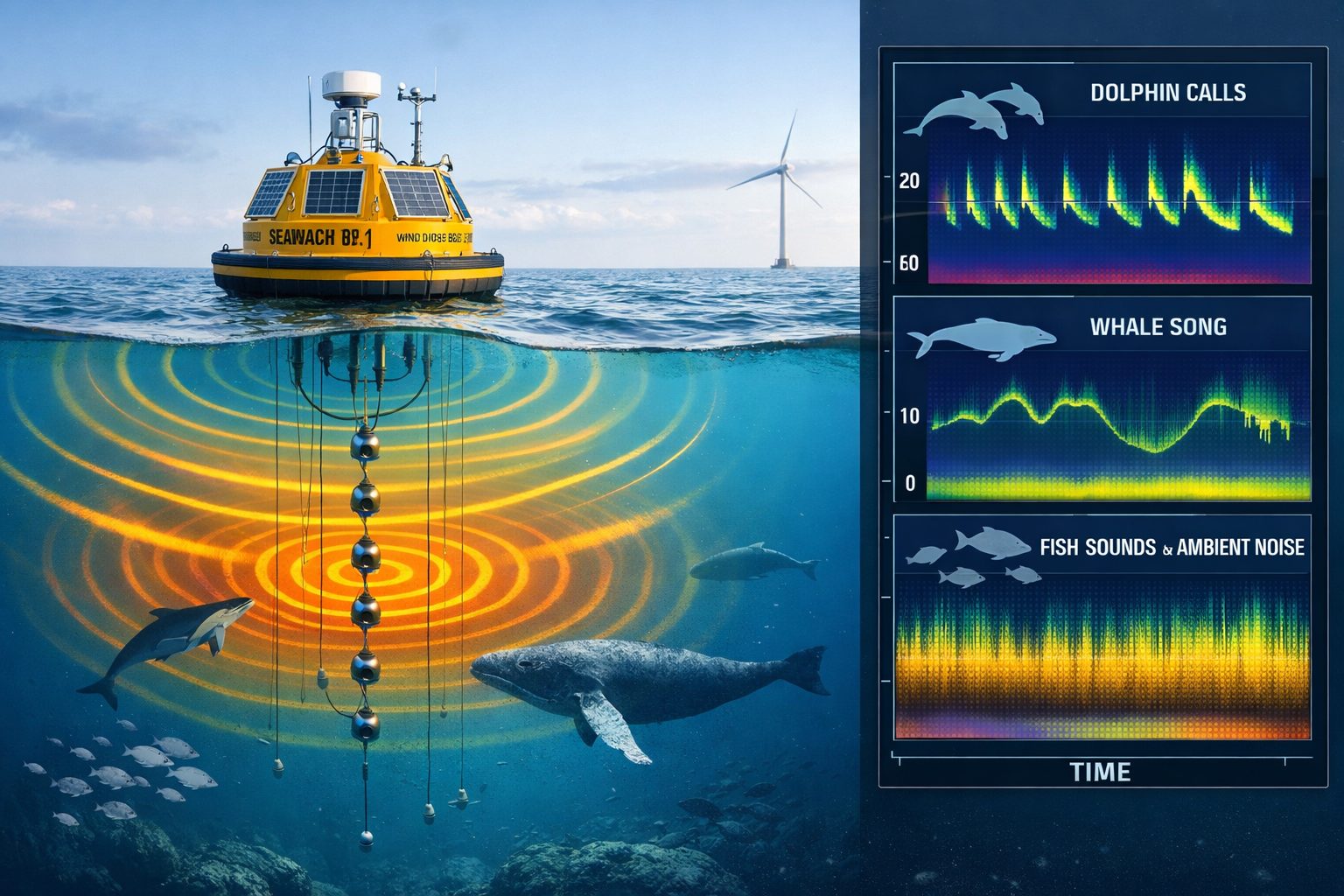
The Synergy of Complementary Methods
The true power of the Radical Transformation in Biodiversity Measurement: eDNA and Acoustic Protocols for 2026 Ecology Surveys emerges when these technologies are deployed together. Integration of bioacoustics and eDNA represents current best practice for holistic biodiversity monitoring, combining continuous acoustic soundscape analysis with genetic detection to answer complementary questions about species presence, temporal patterns, and habitat distribution.[1]
This combined approach offers distinct advantages:
| Method | Primary Strengths | Best Applications | Limitations |
|---|---|---|---|
| eDNA | Broad taxonomic coverage, detects elusive/rare species, minimal disturbance | Species inventories, invasive species detection, aquatic biodiversity | Limited abundance quantification, reference database gaps |
| Bioacoustics | Temporal patterns, behavioral data, continuous monitoring | Activity patterns, breeding events, habitat use | Limited to vocal species, requires acoustic reference libraries |
| Combined | Comprehensive detection, temporal + spatial data, validation cross-check | Complete baseline assessments, long-term monitoring, BNG verification | Higher initial investment, specialized analytical expertise |
Practical Implementation for Development Projects
For developers and land managers seeking to achieve 10% biodiversity net gain, implementing these protocols requires strategic planning:
Phase 1: Baseline Assessment (Pre-Development)
- Deploy acoustic recording units across representative habitat types
- Collect eDNA samples from all water bodies, soil types, and microhabitats
- Establish sampling schedules that capture seasonal variation (minimum 12 months recommended)
- Integrate data with traditional survey methods for validation
Phase 2: Construction Monitoring
- Maintain acoustic monitoring to detect disturbance impacts
- Collect periodic eDNA samples to track species persistence
- Document changes in species composition and activity patterns
- Adjust mitigation measures based on real-time data
Phase 3: Post-Development Verification
- Resume intensive monitoring protocols to assess recovery
- Compare species diversity and abundance to baseline conditions
- Generate evidence for biodiversity impact assessments
- Support long-term habitat management and adaptive strategies
eDNA Expeditions: Global Standardization Initiative
The eDNA Expeditions 2026-2028 initiative represents an unprecedented effort to establish global standards and capacity for molecular biodiversity monitoring. This program encompasses:
- 25 marine sites across diverse biogeographic regions
- Capacity development in biomolecular sampling techniques
- Community involvement protocols that engage local stakeholders
- "Tree-of-life" bioinformatic workflows detecting biodiversity from microbes to megafauna[2]
The timeline for this transformative initiative includes:
- Q4 2026 – Q2 2027: First marine sampling campaigns begin
- Q3 2027 – Q4 2028: Comprehensive data integration and analysis
- 2028: Global publication and expansion planning[2]
Critically, all eDNA Expeditions data will be openly shared through OBIS (Ocean Biodiversity Information System) following FAIR principles (Findable, Accessible, Interoperable, Reusable). Site-specific curated species lists and interactive dashboards will be delivered directly to participating locations, supporting local monitoring and management priorities.[2]
Addressing Current Limitations and Challenges
Despite remarkable advances, several challenges require ongoing attention:
Reference Database Gaps 🧬
- Many species lack genetic reference sequences in public databases
- Taxonomic identification depends on comprehensive reference libraries
- Ongoing sequencing efforts continue expanding coverage
Abundance Quantification 📊
- eDNA detection indicates presence but doesn't directly measure population size
- Environmental factors affect DNA degradation and detection probability
- Quantitative PCR methods and modeling approaches are improving abundance estimates[3]
Technical Expertise Requirements 🔬
- Laboratory analysis requires specialized equipment and training
- Bioinformatic processing demands computational resources
- Field sampling protocols need careful quality control
Cost Considerations 💰
- Initial equipment investment can be substantial
- Laboratory analysis costs vary by taxonomic scope and sample number
- Long-term monitoring requires sustained funding commitments
For developers evaluating biodiversity unit costs and project budgets, these factors must be weighed against the comprehensive data quality and reduced field time that molecular methods provide.
Supporting Biodiversity Net Gain and Conservation Frameworks
The Radical Transformation in Biodiversity Measurement: eDNA and Acoustic Protocols for 2026 Ecology Surveys directly supports multiple regulatory and conservation frameworks:
Biodiversity Net Gain Compliance
Combined bioacoustics and eDNA approaches strengthen environmental baselines and enable faster, more reliable change detection.[1] This capability is essential for:
- Establishing accurate pre-development biodiversity metrics
- Monitoring habitat creation and enhancement effectiveness
- Documenting species colonization of created habitats
- Verifying on-site or off-site delivery outcomes
- Supporting biodiversity credit trading with robust evidence
International Conservation Targets
These monitoring protocols align with and support:
- Marine Strategy Framework Directive (MSFD): Comprehensive marine ecosystem assessment
- Kunming-Montreal Global Biodiversity Framework: 30×30 conservation targets and biodiversity mainstreaming
- UK Biodiversity Action Plans: Species and habitat monitoring requirements
- Marine spatial planning: Evidence-based zoning and protection measures[1]
Citizen Science Integration
An exciting dimension of this transformation involves citizen science participation. Simplified eDNA collection protocols allow trained volunteers to contribute to large-scale monitoring efforts:
- Community members can collect water samples following standardized protocols
- Local knowledge enhances site selection and temporal sampling strategies
- Engagement builds public support for conservation initiatives
- Cost-effective expansion of monitoring coverage across landscapes
This democratization of biodiversity monitoring aligns with recommendations for revolutionizing BNG accuracy in dynamic ecosystems through broader participation and more frequent sampling.
Future Directions and Emerging Technologies
The field continues to evolve rapidly, with several promising developments on the horizon:
Portable Sequencing Devices 📱
- Handheld DNA sequencers enable field-based species identification
- Real-time results inform adaptive management decisions
- Reduced laboratory dependency and faster turnaround times
Artificial Intelligence Integration 🤖
- Machine learning algorithms automate acoustic species identification
- AI-powered bioinformatic pipelines accelerate data processing
- Pattern recognition identifies ecosystem changes and anomalies
Multi-Kingdom Detection 🌿
- Simultaneous detection of plants, animals, fungi, and microorganisms
- Ecosystem-level assessment from single sample sets
- Understanding of trophic interactions and ecological networks
Quantitative Advances 📈
- Improved abundance estimation from eDNA concentrations
- Integration with population modeling frameworks
- Predictive capabilities for population trends
For professionals planning biodiversity strategies, staying current with these technological advances ensures projects benefit from the most effective monitoring approaches available.
Conclusion
The Radical Transformation in Biodiversity Measurement: eDNA and Acoustic Protocols for 2026 Ecology Surveys represents far more than technological novelty—it fundamentally reshapes our capacity to understand, monitor, and protect biodiversity in an era of rapid environmental change. By combining the broad taxonomic coverage and elusive species detection of eDNA with the temporal richness and behavioral insights of bioacoustic monitoring, ecology professionals can now generate comprehensive ecosystem assessments that were simply impossible just a few years ago.
For developers, land managers, and conservation practitioners, these protocols offer practical pathways to:
✅ Establish robust environmental baselines that capture true biodiversity rather than just conspicuous species
✅ Monitor project impacts with unprecedented sensitivity and temporal resolution
✅ Demonstrate biodiversity net gain with compelling, quantitative evidence
✅ Reduce survey costs and timelines through automation and efficient sampling
✅ Support adaptive management with real-time data that informs decision-making
Actionable Next Steps
Ready to implement these transformative protocols in your projects? Consider these concrete actions:
-
Assess Current Survey Approaches: Evaluate whether traditional methods are capturing complete biodiversity profiles for your sites
-
Engage Specialist Consultants: Work with experienced biodiversity surveyors who can design integrated eDNA and acoustic monitoring programs
-
Plan for Seasonal Coverage: Budget for year-round sampling that captures temporal variation critical for robust baselines
-
Invest in Baseline Quality: Comprehensive pre-development data pays dividends throughout project lifecycle and regulatory processes
-
Explore Automated Technologies: Consider automated samplers and acoustic recorders for long-term monitoring efficiency
-
Stay Informed: Follow developments in eDNA Expeditions and emerging standardization efforts that may affect best practices
-
Integrate with BNG Planning: Ensure molecular and acoustic data feeds directly into biodiversity net gain assessments and compliance documentation
The convergence of molecular biology, acoustic technology, and ecological science is revolutionizing biodiversity measurement in 2026. Organizations that embrace these protocols position themselves at the forefront of environmental stewardship, regulatory compliance, and conservation innovation. The question is no longer whether to adopt these transformative approaches, but how quickly you can integrate them into your biodiversity strategy.
Contact biodiversity specialists today to explore how eDNA and acoustic protocols can strengthen your project's environmental performance and biodiversity outcomes. 🌱
References
[1] Holistic Biodiversity Monitoring Combining Bioacoustics And Edna To Deepen Our Understanding Of Marine Life – https://www.fugro.com/news/long-reads/2026/holistic-biodiversity-monitoring-combining-bioacoustics-and-edna-to-deepen-our-understanding-of-marine-life
[2] ednaexpeditions – https://ednaexpeditions.org
[3] pubmed.ncbi.nlm.nih.gov – https://pubmed.ncbi.nlm.nih.gov/41522225/
[4] Edn3 – https://onlinelibrary.wiley.com/doi/10.1002/edn3.70222


