The Japanese beetle (Popillia japonica) was detected in Swiss vineyards several weeks before a single insect became visible to trained surveyors—not through sharper human eyes, but via autonomous drones collecting environmental DNA from leaf surfaces. This temporal advantage represents a fundamental shift in how conservation professionals establish biodiversity baselines, particularly as regulatory frameworks like the UK's Biodiversity Net Gain requirements demand unprecedented accuracy in documenting species presence before development begins.
Invasive Species Early Detection Networks: Drone and eDNA Strategies for Biodiversity Baseline Accuracy have emerged as critical tools in 2026, combining aerial surveillance technology with molecular detection methods to identify biological threats before they establish reproducing populations. This integration directly impacts the reliability of biodiversity assessments that form the foundation of conservation planning and regulatory compliance.

Key Takeaways
- Temporal advantage: eDNA detection identifies invasive species 3-4 weeks earlier than conventional visual surveys, enabling intervention before population establishment [1][2]
- Autonomous operation: Fully autonomous drone-eDNA systems now operate across multiple European regions without human teleoperation, dramatically reducing monitoring costs [2]
- Multi-species capability: Integrated networks detect diverse biological threats simultaneously using metabarcoding combined with species-specific confirmation protocols [1][4]
- Baseline accuracy improvement: Early detection networks eliminate false-negative errors in pre-development surveys, strengthening Biodiversity Net Gain compliance and reducing regulatory risk
- Operational workflow established: Evidence-based management sequences—eDNA flagging → camera confirmation → density measurement → removal verification—provide replicable frameworks for conservation practitioners [1]
Understanding Invasive Species Early Detection Networks
Early detection networks represent coordinated surveillance systems designed to identify non-native species at the earliest possible stage of invasion, when populations remain small, localized, and manageable. Unlike reactive monitoring that responds after establishment, these networks deploy multiple detection technologies across high-risk entry points and vulnerable ecosystems to intercept biological threats during the critical window when eradication remains feasible.
The Detection Timeline Challenge
Traditional biodiversity surveys rely on visual observation, trapping, or acoustic monitoring—methods that require sufficient population density to generate detectable signals. A single Japanese beetle in a 10-hectare vineyard produces no visual signature. Ten beetles scattered across the same area remain effectively invisible. Only when populations reach hundreds or thousands of individuals do conventional methods reliably detect presence.
This detection lag creates a fundamental problem for baseline assessments. A site surveyed in spring may appear free of invasive species, yet harbor low-density populations that explode by autumn. Development proceeds based on inaccurate baseline data, triggering compliance failures when post-construction monitoring reveals species previously "absent."
Three-Tier Detection Architecture
Modern early detection networks operate through three complementary surveillance tiers:
Tier 1: High-Risk Entry Point Monitoring 🚨
Concentrated surveillance at locations where invasive species most likely arrive—ports, agricultural import facilities, irrigation canal inlets, boat ramps, and nurseries. These sites receive frequent eDNA sampling and visual inspection.
Tier 2: Regional Grid Surveillance 🗺️
Systematic sampling across broader landscapes using standardized protocols. Drone-eDNA systems excel at this tier, covering large areas with consistent methodology that human surveyors cannot match economically.
Tier 3: Targeted Response Verification 🎯
Intensive follow-up monitoring triggered by positive detections, combining eDNA, camera traps, physical surveys, and population density assessments to confirm establishment status and guide management decisions.
This architecture ensures that biodiversity baseline documentation captures species presence even at densities below traditional detection thresholds.
Drone Technology for Invasive Species Surveillance
Autonomous aerial systems have evolved from experimental platforms to operational tools deployed across multiple European regions in 2026. The ETH Zurich project completed in early 2026 demonstrated fully autonomous (not remotely piloted) eDNA sampling conducted by drones across Swiss viticulture and broader regional applications [2].
Autonomous vs. Teleoperated Systems
Autonomous systems operate without real-time human control, following pre-programmed flight paths, making sampling decisions based on onboard sensors, and adapting to environmental conditions through machine learning algorithms. These platforms reduce operational costs by 60-75% compared to piloted alternatives while maintaining consistent sampling protocols that eliminate human variability [2].
Key capabilities of 2026 autonomous drone-eDNA platforms include:
- GPS-guided systematic sampling across predetermined grid patterns
- Adaptive flight path adjustment responding to wind, precipitation, and obstacle detection
- Automated sample collection using robotic arms or vacuum systems
- Onboard sample preservation maintaining DNA integrity during multi-hour missions
- Multi-sensor integration combining eDNA collection with thermal imaging, multispectral cameras, and LiDAR for habitat characterization
Coverage Efficiency and Cost Analysis
A single autonomous drone system can survey 200-300 hectares per day with systematic eDNA sampling—equivalent to 15-20 human survey teams working simultaneously. Cost-benefit analysis demonstrates that eDNA rapid tests reduce financial and temporal costs for localities while providing accurate invasive species detection [3].
| Monitoring Method | Daily Coverage | Cost per Hectare | Detection Sensitivity | Weather Dependency |
|---|---|---|---|---|
| Visual Survey Teams | 5-8 hectares | £45-£65 | Low (dense populations only) | High |
| Conventional Drone (Piloted) | 50-80 hectares | £12-£18 | Medium (visual identification) | Medium |
| Autonomous Drone-eDNA | 200-300 hectares | £3-£6 | High (single organisms) | Low |
This efficiency transformation enables developers to conduct comprehensive pre-construction surveys across entire project sites rather than sampling representative plots—a critical improvement for achieving Biodiversity Net Gain compliance with defensible baseline data.
Multi-Species Detection in Agricultural Settings
The ETH Zurich project successfully detected both Popillia japonica (Japanese beetle) and Scaphoideus titanus (grapevine leafhopper vector), establishing protocol transferability for monitoring diverse biological threats [2]. This multi-species capability means a single drone mission generates detection data for dozens of target organisms simultaneously—impossible with species-specific visual surveys or trap systems.
Agricultural applications extend beyond pest management to invasive plant detection, pollinator monitoring, and soil microbiome assessment—all relevant to biodiversity planning for development projects near agricultural land.
Environmental DNA (eDNA) Sampling and Analysis Strategies
Environmental DNA refers to genetic material organisms shed into their surroundings through skin cells, scales, feces, urine, gametes, and decomposing tissue. Water, soil, air, and surfaces accumulate this molecular signature, creating a detection medium that persists for days to weeks depending on environmental conditions.

Collection Methods Across Ecosystem Types
Aquatic environments remain the most established eDNA application, with standardized protocols for water filtration through 0.45-micron membranes that capture cellular material. High sensitivity for identifying rare, endangered, and invasive species has been confirmed across aquatic ecosystems, including successful detection of invasive crayfish in Europe, red drum in the East China Sea, and New Zealand mud snails [4].
Terrestrial applications collect eDNA from:
- Soil cores capturing DNA from burrowing organisms, root systems, and decomposing material
- Surface swabs sampling leaf surfaces, tree bark, and rock faces where organisms contact or rest
- Air filtration capturing pollen, fungal spores, and insect fragments suspended in atmosphere
- Passive collectors using adhesive surfaces or water traps that accumulate genetic material over time
Atmospheric eDNA, the newest frontier, has detected Argentine ants and other terrestrial invasive species through air sampling [4]—particularly relevant for flying insects that traditional ground-based methods struggle to monitor.
Two-Tiered Analysis: Metabarcoding and Species-Specific qPCR
Many agencies now combine metabarcoding (broad scouting) with species-specific quantitative PCR (targeted confirmation) rather than relying on single-method approaches, improving accuracy and management credibility [1].
Metabarcoding workflow:
- Extract DNA from environmental sample
- Amplify conserved genetic regions (COI, 16S, ITS) present across many species
- Sequence amplified DNA using high-throughput platforms
- Compare sequences against reference databases
- Generate species inventory from matches
Advantages: Detects unexpected species, provides community composition data, identifies multiple invasive threats simultaneously
Limitations: Lower sensitivity for individual species, requires comprehensive reference databases, higher per-sample cost
Species-specific qPCR workflow:
- Extract DNA from environmental sample
- Amplify unique genetic marker for target species only
- Measure amplification in real-time to quantify DNA concentration
- Compare against standard curves and negative controls
- Confirm presence/absence with statistical confidence
Advantages: Extremely high sensitivity, lower per-sample cost, rapid results (4-6 hours), established validation protocols
Limitations: Detects only pre-selected target species, misses unexpected invasions
Workflow Optimization and Result Delivery
Technical advancements during the ETH Zurich project's final phase included optimization of DNA workflows, which substantially reduced the time required for result delivery—critical for rapid-response management decisions [2]. Standard laboratory processing now achieves:
- Field to extraction: 24-48 hours with proper sample preservation
- Extraction to qPCR results: 4-8 hours for species-specific assays
- Extraction to metabarcoding results: 3-5 days including sequencing and bioinformatics
- Total detection timeline: 2-7 days from sample collection to confirmed identification
This timeline enables weekly surveillance cycles that track invasive species spread in near-real-time—transforming reactive management into predictive intervention.
Operational Limitations and Complementary Verification Methods
eDNA technology has clear limitations for enforcement decisions: eDNA cannot determine how many individuals are present, whether they are reproducing, where exactly the organism is located, or whether the population is established or transient—limiting its use as standalone proof for enforcement or eradication without follow-up monitoring [1].
What eDNA Cannot Tell You
Population size: DNA concentration correlates poorly with organism abundance due to variable shedding rates, environmental degradation, and transport dynamics. A single dead organism may produce stronger eDNA signal than ten living individuals.
Reproductive status: Detecting species DNA confirms presence but provides no information about breeding success, viable populations, or establishment trajectory.
Precise location: Aquatic eDNA travels downstream; atmospheric eDNA disperses with wind. Detection indicates presence within a watershed or airshed, not specific coordinates.
Living vs. dead: DNA persists after organism death, creating false-positive risks in areas where recent eradication occurred or where organisms passed through without establishing.
Integrated Workflow for Evidence-Based Management
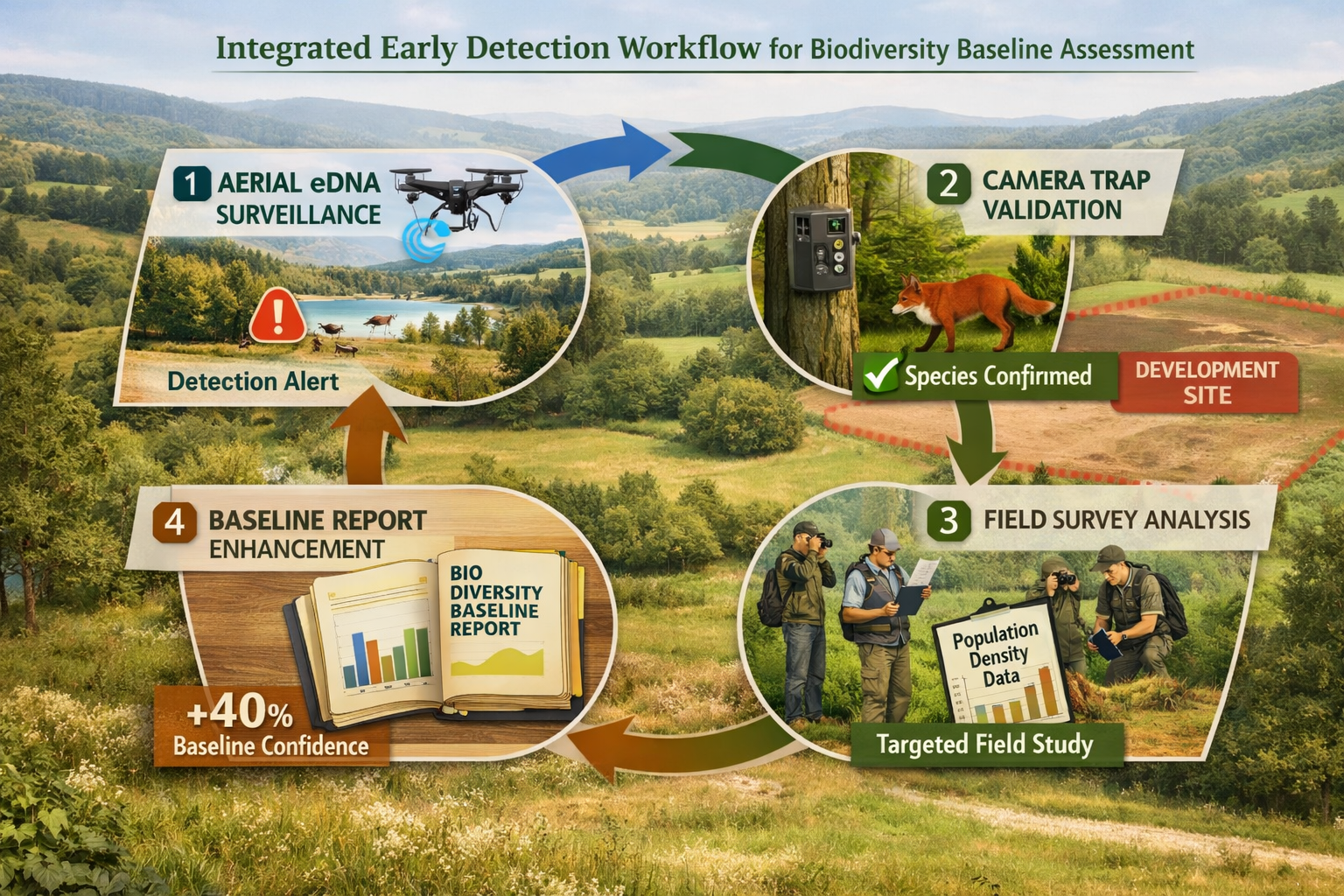
The integrated drone-camera-eDNA workflow emerging as best practice follows a defined sequence: eDNA detection flags a zone of concern → camera deployment confirms species identity → targeted trapping measures density → follow-up eDNA verifies removal success → expansion to new zones, combining strengths of each technology [1].
Stage 1: eDNA Surveillance (Detection)
Systematic drone-eDNA sampling across entire project site generates presence/absence data for target invasive species list. Positive detections trigger Stage 2.
Stage 2: Camera Trap Verification (Confirmation)
Deployment of motion-activated cameras or visual inspection in flagged zones confirms species identity and provides photographic evidence. Multiple independent observations establish confidence.
Stage 3: Population Assessment (Quantification)
Targeted trapping, mark-recapture studies, or intensive visual surveys estimate population size, distribution, and demographic structure. This stage determines management feasibility.
Stage 4: Removal and Verification (Validation)
Management intervention followed by repeated eDNA sampling confirms eradication success or detects residual/reinvading populations requiring additional treatment.
This four-stage protocol provides defensible documentation for regulatory compliance while avoiding the false-positive and false-negative errors that compromise single-method approaches.
Three Primary Operational Applications in Conservation Planning
Agencies currently deploy eDNA for early detection at low-density populations, surveillance of high-risk entry points (farm ponds, boat ramps, irrigation canals, tributaries), and post-removal verification to confirm eradication success or detect reinvasion [1].
Application 1: Pre-Development Baseline Surveys
Challenge: Traditional baseline surveys conducted over 1-2 seasons may miss invasive species at low density, creating liability when post-construction monitoring reveals previously "undetected" populations.
Solution: Drone-eDNA surveillance across entire development footprint generates comprehensive species inventory with sensitivity to single organisms, eliminating false-negative errors that undermine Biodiversity Net Gain assessments.
Implementation: Quarterly drone-eDNA surveys throughout pre-construction period, with metabarcoding to detect unexpected species and species-specific qPCR for known regional invasive threats. Results integrate with habitat surveys to produce comprehensive baseline documentation.
Application 2: High-Risk Entry Point Monitoring
Challenge: Invasive species enter through predictable pathways—imported materials, contaminated equipment, watercraft, and agricultural products—but monitoring every potential entry point exceeds available resources.
Solution: Permanent or semi-permanent eDNA sampling stations at high-risk locations provide continuous surveillance that flags introductions within days of arrival, enabling rapid response before establishment.
Implementation: Weekly water sampling at irrigation inlets, boat ramps, and stormwater outfalls; monthly soil sampling at material staging areas; quarterly atmospheric sampling near import facilities. Automated analysis pipelines generate alerts when target species DNA detected.
Application 3: Post-Management Verification
Challenge: Declaring eradication success requires proving absence—statistically impossible through visual surveys that may simply miss remaining individuals.
Solution: Intensive eDNA sampling provides higher confidence in absence declarations, with detection probability curves quantifying surveillance effort required to achieve specified confidence levels (typically 95% confidence of detection if species present at target density).
Implementation: Following invasive species removal, conduct monthly eDNA surveillance for 12-24 months across treatment area and surrounding buffer zones. Continued negative results combined with visual surveys support eradication declaration; positive detections trigger renewed treatment.
Invasive Species Early Detection Networks and Biodiversity Baseline Design
The integration of drone and eDNA strategies fundamentally changes how conservation professionals approach baseline documentation for Biodiversity Net Gain compliance. Traditional habitat-based assessments assume that surveyed species lists represent actual site composition—an assumption that early detection networks reveal as frequently incorrect.
Accuracy Improvements in Baseline Documentation
False-negative elimination: eDNA detection of invasive species at densities below visual detection thresholds prevents the baseline error where sites are incorrectly classified as "invasive-free," then require corrective action post-construction when populations become apparent.
Temporal coverage extension: Drone-eDNA systems operate in weather conditions and seasons when human surveyors cannot work effectively, extending survey windows and capturing species with brief activity periods or nocturnal behavior.
Spatial coverage completeness: Autonomous platforms survey entire project footprints rather than accessible representative plots, eliminating sampling bias toward easily reached areas while missing difficult terrain where invasive species often establish.
Reproducibility and audit trail: Automated systems generate GPS-tagged samples with standardized protocols, creating defensible documentation that regulatory agencies can verify and independent auditors can validate.
Risk Reduction for Development Projects
Projects that establish baselines using integrated detection networks reduce several categories of regulatory and financial risk:
Compliance risk: Accurate baseline data prevents disputes over whether invasive species were present pre-construction or arrived post-development—a common source of Biodiversity Net Gain enforcement actions.
Remediation cost risk: Early detection enables low-cost intervention before populations establish, avoiding expensive eradication programs that may cost 10-100× more than early-stage removal.
Timeline risk: Discovering invasive species mid-construction triggers work stoppages, regulatory review, and management plans that delay project completion. Pre-construction detection allows planned mitigation without schedule disruption.
Reputation risk: High-profile invasive species spread from development sites generates negative publicity and stakeholder opposition. Proactive detection demonstrates environmental stewardship and due diligence.
Global Standardization and Policy Integration
Global standardization efforts are underway through coordinated initiatives including the 2026 Global eDNA Conference and GuardIAS eDNA Summer School (June 25–July 7, 2026), which focus on developing standardized sampling methods, data management protocols, and best practices for global adoption of eDNA surveillance networks [5][6].
Emerging International Standards
Sampling protocols: Standardized methods for water volume, filtration pore size, filter material, preservation buffers, and storage conditions ensure comparability across laboratories and regions.
Quality assurance/quality control: Required negative controls (field blanks, extraction blanks, PCR blanks) and positive controls (synthetic DNA standards, reference samples) with documented acceptance criteria.
Reference database requirements: Minimum standards for genetic reference libraries including voucher specimen documentation, sequence quality metrics, and taxonomic verification by qualified experts.
Data management: Standardized metadata reporting including GPS coordinates, environmental conditions, sampling method, extraction protocol, amplification primers, and bioinformatics pipelines—enabling data sharing and meta-analysis.
Policy Framework Development
Emerging conference and training session focus areas include scaling eDNA into global policy frameworks, biosecurity applications, Indigenous partnerships, and community science engagement—indicating institutional adoption moving beyond research phase [6].
Biosecurity regulations: Several jurisdictions now accept eDNA evidence for quarantine decisions, import restrictions, and eradication declarations when protocols meet specified quality standards.
Conservation status assessments: Endangered species listings increasingly incorporate eDNA survey data to document range contractions, habitat use, and population persistence.
Environmental impact assessment: Regulatory agencies require or recommend eDNA baseline surveys for projects in sensitive ecosystems or regions with high invasive species risk.
Biodiversity Net Gain: UK guidance increasingly references molecular detection methods as best practice for establishing accurate baselines, particularly for aquatic and soil-dwelling species difficult to survey conventionally.
Implementation Considerations for Conservation Practitioners
Organizations implementing Invasive Species Early Detection Networks: Drone and eDNA Strategies for Biodiversity Baseline Accuracy should consider several practical factors that determine program success.
Budget and Resource Allocation
Initial investment: Autonomous drone systems cost £15,000-£45,000 depending on capabilities; eDNA laboratory setup requires £25,000-£75,000 for extraction and amplification equipment. Alternatively, contract services eliminate capital costs.
Operational costs: Per-sample eDNA analysis ranges from £15-£40 for species-specific qPCR to £80-£150 for metabarcoding. Drone operation costs £200-£400 per day including operator, maintenance, and insurance.
Cost-benefit threshold: Programs become cost-effective when monitoring requirements exceed approximately 500 hectares annually or when early detection prevents single eradication program costing more than 3-year surveillance investment [3].
Technical Capacity Requirements
Personnel training: Drone pilots require Civil Aviation Authority certification; eDNA sampling teams need contamination prevention training; laboratory staff require molecular biology expertise.
Quality management: Successful programs implement documented standard operating procedures, proficiency testing, and regular audits to maintain data quality and regulatory acceptance.
Data management infrastructure: GPS-tagged samples, laboratory results, and environmental metadata require database systems that support spatial analysis, temporal trending, and regulatory reporting.
Partnership and Collaboration Models
Academic partnerships: Universities provide access to advanced sequencing platforms, bioinformatics expertise, and research support that reduces implementation costs while advancing methodology.
Agency coordination: Regional surveillance networks achieve greatest efficiency when multiple agencies share data, coordinate sampling to avoid duplication, and jointly fund infrastructure.
Citizen science integration: Trained volunteers can collect eDNA samples following standardized protocols, dramatically expanding surveillance coverage while building public engagement and support.
Future Developments and Emerging Technologies
The field continues rapid evolution with several emerging capabilities that will further enhance early detection networks:
Portable sequencing platforms: Handheld DNA sequencers enable field-based species identification within hours, eliminating laboratory delays and enabling real-time management decisions during survey operations.
Machine learning integration: AI algorithms trained on multispectral imagery, thermal signatures, and habitat characteristics predict high-risk invasion zones, optimizing surveillance effort allocation.
Swarm robotics: Coordinated fleets of autonomous drones with distributed sampling create dense spatial coverage and adaptive sampling that concentrates effort where detections occur.
Quantitative eDNA: Improved understanding of DNA shedding rates, degradation kinetics, and transport dynamics enables population size estimation from DNA concentration—addressing current limitation.
Multi-kingdom detection: Simultaneous detection of invasive plants, animals, fungi, and pathogens from single environmental sample using universal primers and comprehensive reference databases.
Conclusion
Invasive Species Early Detection Networks: Drone and eDNA Strategies for Biodiversity Baseline Accuracy represent a fundamental advancement in conservation practice, providing detection capabilities that identify biological threats weeks before conventional methods while covering spatial scales impossible for traditional survey approaches. The 2026 operational deployment of fully autonomous drone-eDNA systems across European agricultural regions demonstrates that these technologies have matured beyond research applications into reliable tools for routine biodiversity monitoring.
For organizations implementing Biodiversity Net Gain requirements, the integration of early detection networks into baseline assessments eliminates false-negative errors that create regulatory compliance risk and financial liability. The temporal advantage of eDNA detection—identifying species 3-4 weeks before visual observation—enables intervention during the critical window when populations remain manageable and eradication remains feasible.
Actionable Next Steps
For developers and planners: Incorporate drone-eDNA baseline surveys into project timelines, particularly for sites near high-risk entry points or in regions with documented invasive species pressure. Early investment in comprehensive baseline documentation reduces downstream compliance risk and demonstrates environmental due diligence.
For conservation practitioners: Establish partnerships with eDNA laboratories and certified drone operators to build capacity for integrated surveillance. Begin with pilot programs targeting 2-3 high-priority invasive species before expanding to comprehensive monitoring networks.
For landowners: Consider participating in regional surveillance networks that provide early warning of invasive species arrival, enabling proactive management before populations establish. Biodiversity unit sales may benefit from documented invasive species management programs that protect habitat quality.
For regulatory agencies: Develop guidance documents specifying quality standards for eDNA evidence in baseline assessments, enforcement decisions, and eradication declarations. Support standardization efforts that enable data sharing across jurisdictions and programs.
The convergence of autonomous aerial systems, molecular detection methods, and standardized protocols creates unprecedented opportunity to shift from reactive invasive species management to predictive intervention. Organizations that implement these technologies in 2026 position themselves at the forefront of evidence-based conservation practice while building competitive advantage in an increasingly rigorous regulatory environment.
References
[1] Edna Testing For Invasive Species – https://landwildlifereport.com/2026/01/20/edna-testing-for-invasive-species/
[2] Autonomous Edna Surveillance For Pest Management – https://worldfoodsystem.ethz.ch/news/wfsc-news/2026/01/autonomous-edna-surveillance-for-pest-management.html
[3] A Cost Benefit Analysis Of Edna Technology For Invasive Species Detection – https://lafollette.wisc.edu/research/a-cost-benefit-analysis-of-edna-technology-for-invasive-species-detection/
[4] Pmc12789655 – https://pmc.ncbi.nlm.nih.gov/articles/PMC12789655/
[5] Guardias Edna Summer School 2026 – https://www.sednasociety.com/post/guardias-edna-summer-school-2026
[6] Global Edna Conference – https://www.mtsociety.org/global-edna-conference


