Biodiversity Net Gain (BNG) assessments in 2026 measure visible species—birds, plants, insects—but overlook the invisible architects of ecosystem health: microbial communities. Recent research demonstrates that holobiont approaches, which examine organisms alongside their symbiotic microbiomes, provide "holistic insight into the range of ecological interactions between microbes and other organisms" and can transform environmental management and conservation strategies[3]. As mandatory BNG legislation now extends to nationally significant infrastructure projects requiring 10% biodiversity gains with 30-year monitoring periods[1], the question emerges: how can ecologists integrate Holobiont Surveys in BNG Projects: Protocols for Assessing Host-Microbe Symbioses in Restored Ecosystems to capture the full complexity of ecological restoration success?

Key Takeaways
- Holobiont surveys examine organisms plus their microbial partners, revealing hidden symbiotic relationships critical to ecosystem function
- Environmental DNA/RNA (eNA) methods enable non-invasive assessment of microbial communities in restored habitats without extensive culturing
- BNG projects must now demonstrate measurable biodiversity gains over 30 years, creating opportunities for microbial monitoring protocols
- Competent ecologists can integrate holobiont assessments into standard BNG workflows using accessible eDNA sampling techniques
- Baseline microbial surveys establish reference conditions for tracking restoration trajectories and ecosystem recovery
Understanding the Holobiont Concept in Ecological Restoration
The term "holobiont" describes a biological unit comprising a host organism and all its associated microorganisms—bacteria, fungi, archaea, viruses, and protists. This concept fundamentally challenges traditional biodiversity assessments that treat species as isolated entities.
Why Holobionts Matter for BNG Success
In restored ecosystems, visible biodiversity represents only a fraction of functional recovery. Below-ground microbial networks:
- Facilitate nutrient cycling through decomposition and mineralization
- Support plant establishment via mycorrhizal associations
- Enhance soil structure through biofilm formation
- Regulate disease through competitive exclusion of pathogens
- Drive carbon sequestration in soil organic matter
When creating a biodiversity plan for development projects, developers typically focus on habitat area and species richness. However, a woodland planted with native trees may fail to thrive without appropriate ectomycorrhizal fungal partners. A wetland restoration might support waterbirds but lack the methanogenic bacteria essential for biogeochemical cycling.
The Tree of Life Approach to Environmental Assessment
Researchers have begun incorporating Tree of Life frameworks into environmental nucleic acid (eNA) analyses[3]. This approach sequences DNA and RNA from environmental samples to identify the full spectrum of organisms present—from microscopic bacteria to macroscopic fauna.
The advantages for BNG monitoring include:
✅ Non-invasive sampling that doesn't disturb sensitive species
✅ Comprehensive detection of rare and cryptic organisms
✅ Temporal tracking of community assembly during restoration
✅ Cost-effective compared to traditional morphological surveys
✅ Standardized protocols that reduce observer bias
Holobiont Surveys in BNG Projects: Establishing Baseline Protocols
Implementing Holobiont Surveys in BNG Projects: Protocols for Assessing Host-Microbe Symbioses in Restored Ecosystems requires systematic approaches adapted to project scale and habitat type.

Pre-Development Baseline Assessment
Before ground disturbance, competent ecologists—typically evidenced by CIEEM membership[1]—should establish microbial baseline conditions. This creates reference data for measuring net gain.
Recommended sampling strategy:
| Habitat Type | Sample Points | Depth/Location | Replicates |
|---|---|---|---|
| Grassland | 5 per hectare | 0-15cm soil | 3 per point |
| Woodland | 10 per hectare | Rhizosphere + bulk soil | 3 per point |
| Wetland | 8 per hectare | Sediment + water column | 3 per point |
| Hedgerow | 3 per 100m | Root zone | 3 per point |
Environmental DNA Collection Methods
Soil sampling protocol:
- Use sterile corers to collect 10g samples from designated depths
- Composite subsamples within 5m radius to account for spatial heterogeneity
- Store immediately at -20°C or in preservation buffer
- Record GPS coordinates, soil moisture, temperature, and pH
Water sampling for aquatic holobionts:
- Collect 2L surface water in sterile bottles
- Filter through 0.22μm membranes within 6 hours
- Preserve filters in DNA/RNA shield solution
- Document water chemistry parameters (DO, conductivity, nutrients)
Plant-associated microbiome sampling:
- Collect root, leaf, and stem tissues from target species
- Surface sterilize to distinguish endophytes from epiphytes
- Extract both DNA and RNA for active community profiling
- Include multiple individuals across age classes
Laboratory Analysis Workflows
Modern holobiont surveys employ metabarcoding approaches that amplify and sequence specific genetic markers:
16S rRNA gene → Bacterial and archaeal diversity
ITS region → Fungal communities
18S rRNA gene → Eukaryotic microorganisms
COI gene → Metazoan fauna
Bioinformatic pipelines process raw sequence data to generate:
- Operational Taxonomic Units (OTUs) or Amplicon Sequence Variants (ASVs)
- Alpha diversity metrics (species richness, Shannon index)
- Beta diversity analysis (community composition differences)
- Functional gene predictions (metabolic potential)
For BNG projects requiring comprehensive assessment, laboratories should provide taxonomic resolution to genus level where possible, enabling identification of keystone symbiotic taxa.
Integrating Holobiont Metrics into BNG Calculations
Current BNG assessments use the Biodiversity Metric 4.0, which calculates biodiversity units based on habitat distinctiveness, condition, and strategic significance. Holobiont data can enhance condition assessments and provide novel indicators of restoration success.
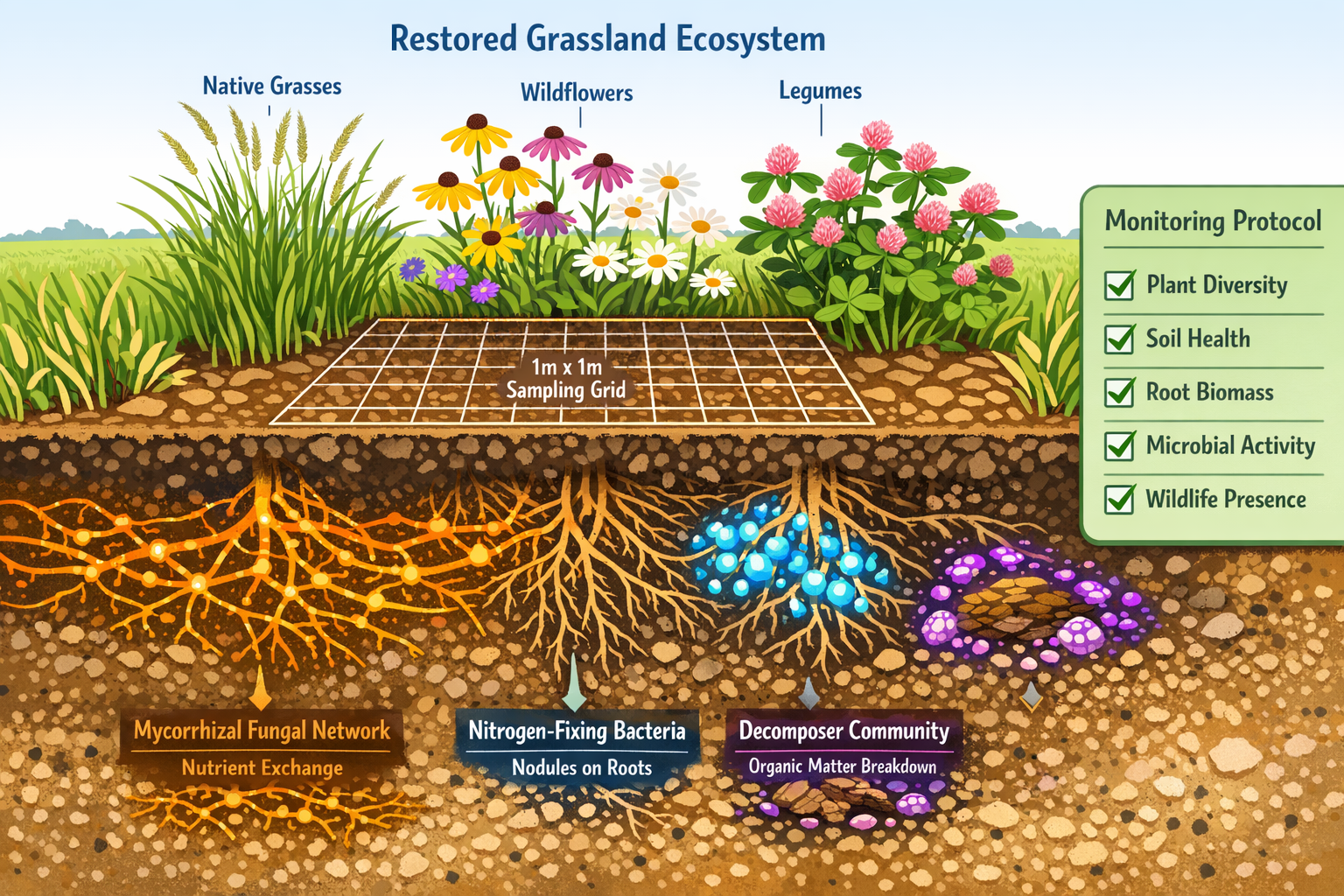
Proposed Holobiont Indicators for BNG Monitoring
Indicator 1: Symbiotic Richness Index
Quantifies the diversity of known beneficial symbioses present in restored habitats. For example:
- Mycorrhizal fungal species associated with planted trees
- Nitrogen-fixing bacteria in legume root nodules
- Pollinator gut microbiomes supporting colony health
Indicator 2: Microbial Community Recovery Trajectory
Compares restored site microbiomes to reference ecosystems using Bray-Curtis dissimilarity. Successful restoration shows convergence toward reference conditions over the 30-year monitoring period[1].
Indicator 3: Functional Gene Abundance
Measures genes involved in ecosystem services:
- Carbon cycling (cellulose degradation, methanogenesis)
- Nutrient transformation (nitrification, denitrification, phosphate solubilization)
- Stress tolerance (drought resistance, heavy metal tolerance)
Indicator 4: Network Complexity
Analyzes co-occurrence patterns to assess ecological interaction networks. Mature, stable ecosystems exhibit complex microbial networks with high connectivity.
Practical Implementation for Small Development Projects
Small development projects may lack budgets for extensive molecular analyses. Simplified holobiont protocols include:
🔬 Visual assessment of mycorrhizal colonization using root staining
🔬 Soil respiration tests as proxy for microbial activity
🔬 Commercial soil health kits measuring microbial biomass carbon
🔬 Targeted qPCR for specific functional genes rather than full metabarcoding
These approaches provide meaningful data at reduced cost while establishing precedent for microbial monitoring in BNG frameworks.
Holobiont Surveys in BNG Projects: Field Protocols for 2026 and Beyond
As BNG legislation expands in 2026, standardized protocols enable consistent holobiont assessments across projects and practitioners.
Seasonal Sampling Considerations
Microbial communities exhibit temporal dynamics influenced by temperature, moisture, and host phenology. Optimal sampling schedules:
Spring (March-May):
- Captures post-winter community recovery
- Assesses plant-microbe establishment in new plantings
- Monitors amphibian skin microbiomes during breeding
Summer (June-August):
- Peak microbial activity and biomass
- Evaluates stress responses during dry periods
- Samples pollinator-associated microbiomes
Autumn (September-November):
- Documents decomposer community activity
- Assesses fungal fruiting body diversity
- Monitors leaf litter colonization
Winter (December-February):
- Baseline for dormant season communities
- Evaluates cold-adapted microbial populations
- Reduced sampling frequency acceptable
Quality Assurance and Contamination Controls
Environmental DNA work requires stringent controls to prevent false positives:
- Field blanks: Sterile water processed identically to samples
- Extraction blanks: Control for laboratory contamination
- PCR negatives: Verify amplification specificity
- Positive controls: Confirm detection sensitivity
- Replication: Minimum three technical replicates per sample
Data Management and Reporting Standards
Holobiont survey data should integrate with existing BNG documentation. Recommended reporting elements:
📊 Executive summary of microbial diversity findings
📊 Comparison tables showing baseline vs. post-restoration metrics
📊 Visual representations (heatmaps, ordination plots, network diagrams)
📊 Taxonomic inventories of detected symbiotic taxa
📊 Functional predictions related to ecosystem services
📊 Recommendations for adaptive management based on microbial indicators
When planning BNG projects, developers should allocate budget for baseline and periodic holobiont surveys within the 30-year monitoring commitment.
Advanced Techniques: Metagenomics and Metatranscriptomics
While metabarcoding provides taxonomic inventories, advanced "omics" approaches reveal functional capabilities and active processes in restored ecosystems.
Shotgun Metagenomics
Sequences all DNA in environmental samples without PCR amplification bias. Benefits include:
- Genome-resolved metagenomics: Reconstructs complete microbial genomes from environmental samples
- Functional annotation: Identifies specific genes and metabolic pathways
- Strain-level resolution: Detects subtle genetic differences within species
- Viral ecology: Characterizes bacteriophages and their hosts
Metatranscriptomics (Environmental RNA)
Captures actively transcribed genes, revealing which organisms are metabolically active and what functions they perform in real-time. This approach directly addresses the question: "Are beneficial symbioses actually functioning in the restored ecosystem?"
For off-site BNG delivery, metatranscriptomics can verify that purchased biodiversity units represent genuinely functional ecosystems with active microbial processes, not just species presence.
Culturomics: Complementing Molecular Approaches
Despite advances in molecular techniques, cultivation remains valuable for:
- Isolating specific symbionts for experimental validation
- Creating microbial inoculants to accelerate restoration
- Phenotypic characterization of functional traits
- Archiving strains for future research and application
High-throughput culturing methods using diverse media formulations can recover previously "unculturable" microorganisms, expanding the toolkit for holobiont surveys.
Case Study Applications in UK BNG Projects
Woodland Creation with Mycorrhizal Inoculation
A hypothetical 5-hectare woodland creation project in Surrey demonstrates holobiont survey integration:
Baseline: Pre-development agricultural soil showed low fungal diversity dominated by generalist saprotrophs. Ectomycorrhizal fungi (essential for oak and beech establishment) were virtually absent.
Intervention: Native tree planting combined with soil inoculation using mycorrhizal spores from nearby ancient woodland.
Monitoring: Annual eDNA surveys tracked fungal community assembly. By year 3, ectomycorrhizal richness increased 400%, with detection of specialist species (Lactarius, Amanita, Cortinarius). Root colonization rates exceeded 80%, indicating functional symbioses.
BNG outcome: Holobiont data provided evidence of accelerated habitat maturation, supporting higher biodiversity unit calculations based on improved condition scores.
Wetland Restoration with Methanogen Monitoring
A 2-hectare wetland restoration in East Anglia focused on recreating fen conditions:
Baseline: Drained agricultural land harbored aerobic soil communities with negligible anaerobic metabolism.
Intervention: Hydrological restoration raised water tables, creating anoxic conditions.
Monitoring: Quarterly sediment sampling tracked methanogenic archaea using qPCR targeting mcrA genes. Methane flux measurements correlated with archaeal abundance.
BNG outcome: Methanogen establishment verified functional wetland biogeochemistry, distinguishing successful restoration from superficial habitat creation. This provided robust evidence for biodiversity unit calculations.
Challenges and Limitations of Holobiont Surveys
Technical Challenges
Taxonomic reference gaps: Many environmental microorganisms lack cultured representatives, limiting identification accuracy. Reference databases improve continuously but remain incomplete for UK ecosystems.
Functional inference uncertainty: Predicting ecosystem functions from taxonomic data involves assumptions. Not all detected taxa are active, and dormant spores may inflate diversity estimates.
Spatial heterogeneity: Microbial communities vary dramatically across centimeter scales, requiring extensive sampling for representative data.
Practical Constraints
Cost considerations: Comprehensive metagenomic analyses cost £500-2,000 per sample. Small development projects may find full holobiont surveys prohibitively expensive.
Expertise requirements: Bioinformatic analysis demands specialized skills beyond traditional ecological training. Partnerships with molecular ecology laboratories become essential.
Interpretation frameworks: Regulatory guidance for incorporating microbial data into BNG calculations remains undeveloped. Ecologists must establish defensible approaches for translating holobiont metrics into biodiversity units.
Regulatory Considerations
Current BNG legislation does not explicitly require microbial monitoring[1]. However, as understanding of ecosystem functioning advances, regulatory frameworks may evolve to recognize holobiont assessments as best practice for demonstrating genuine biodiversity gains.
Ecologists implementing holobiont surveys in 2026 position themselves as innovators, establishing methodologies that may become standard requirements in future BNG iterations.
Future Directions: Holobiont-Informed Restoration Practice

Microbial Inoculants and Assisted Colonization
Holobiont surveys reveal which symbiotic partners are absent from degraded sites. This knowledge enables targeted interventions:
- Mycorrhizal inoculation of bare-root nursery stock before planting
- Soil transplantation from reference sites to introduce microbial communities
- Bioaugmentation with cultured beneficial strains
- Probiotic treatments for stressed or diseased organisms
These approaches accelerate restoration trajectories, potentially reducing the time required to achieve target biodiversity unit values.
Predictive Modeling of Restoration Outcomes
Machine learning algorithms trained on holobiont survey data can predict restoration success probabilities. By analyzing microbial community composition at early timepoints (years 1-3), models forecast long-term ecosystem development, enabling adaptive management interventions before restoration failure becomes evident.
Integration with Remote Sensing
Combining holobiont surveys with hyperspectral imaging and LiDAR creates multi-scale assessment frameworks. Satellite data captures landscape-level habitat structure, while ground-truthing with microbial surveys validates functional ecosystem properties. This integration supports strategic planning of BNG delivery across regional scales.
Citizen Science Opportunities
Simplified holobiont sampling protocols enable community participation in BNG monitoring. Landowners and local naturalists can collect standardized samples for laboratory analysis, expanding spatial coverage while building public engagement with biodiversity conservation. This approach aligns with broader trends toward democratizing environmental science.
Conclusion: Embracing the Invisible Majority in BNG Assessments
Holobiont Surveys in BNG Projects: Protocols for Assessing Host-Microbe Symbioses in Restored Ecosystems represent a paradigm shift in how ecological restoration is measured and valued. As mandatory BNG requirements extend across development sectors in 2026, the opportunity exists to move beyond counting visible species toward genuinely assessing ecosystem functionality.
Microbial communities—the invisible majority comprising most of Earth's biodiversity—drive the processes that sustain habitats and support macroscopic life. By integrating environmental DNA sampling, metabarcoding analysis, and holobiont-focused interpretation into standard BNG workflows, ecologists can:
✅ Demonstrate measurable restoration success with greater confidence
✅ Identify failing interventions early through microbial indicators
✅ Justify higher biodiversity unit values for functionally mature ecosystems
✅ Contribute to scientific understanding of restoration ecology
✅ Position themselves as leaders in innovative conservation practice
Actionable Next Steps for Ecologists and Developers
- Pilot holobiont surveys on current BNG projects to establish baseline competency and refine protocols
- Partner with molecular ecology laboratories to access technical expertise and analytical capacity
- Document methodologies thoroughly to support regulatory acceptance of microbial data
- Engage with CIEEM and other professional bodies to advocate for holobiont assessment standards
- Allocate appropriate budgets within BNG planning frameworks for microbial monitoring
- Communicate findings effectively to clients, planners, and stakeholders using accessible visualizations
The invisible world of host-microbe symbioses holds the key to understanding whether restored ecosystems truly function or merely appear superficially biodiverse. As the field of environmental nucleic acid analysis matures and costs decrease, holobiont surveys will transition from cutting-edge research to routine practice. Ecologists who embrace these methods now will lead the next generation of BNG assessment, ensuring that biodiversity gains are real, measurable, and sustainable across the required 30-year monitoring periods.
The future of ecological restoration is microbial. The protocols exist. The technology is accessible. The question for 2026 is not whether to incorporate holobiont surveys into BNG projects, but how quickly the industry can adopt these transformative approaches to truly measure what matters: the living, functioning ecosystems we create for future generations.
References
[1] Yorhub Bng Presentation – https://www.yorhub.com/app/uploads/2026/01/YORhub-BNG-Presentation.pdf
[3] pubmed.ncbi.nlm.nih.gov – https://pubmed.ncbi.nlm.nih.gov/39164135/


