Conservation science in 2026 has reached a turning point. As molecular manipulation technologies reshape natural ecosystems—from gene drives controlling invasive species to climate-adapted organisms being introduced into struggling habitats—environmental DNA (eDNA) survey techniques have become essential tools for tracking genetic changes across landscapes. For biodiversity surveyors, developers, and landowners navigating biodiversity net gain requirements, understanding how to detect and monitor these molecular-level shifts has transformed from optional expertise into regulatory necessity.
The Molecular Manipulation Impacts: eDNA Survey Techniques for Genetic Changes in 2026 Ecosystems represent a fundamental evolution in how professionals assess, monitor, and verify ecosystem health. Traditional survey methods—netting, trapping, visual observation—can no longer capture the genetic complexity of modern conservation challenges. This comprehensive guide explores the cutting-edge eDNA protocols now reshaping biodiversity assessment practices.
Key Takeaways
- 🧬 eDNA detection is now faster, cheaper, and more sensitive than traditional survey methods, with Cornell researchers developing predictive models that track where sampled DNA originated in water bodies [1]
- ⏱️ DNA persistence windows standardized at 24-48 hours in most aquatic environments, providing reliable detection timeframes for monitoring genetic changes [4]
- 🔬 Digital droplet PCR (ddPCR) offers superior sensitivity compared to standard qPCR, enabling detection of rare and invasive species at extremely low densities [3]
- 📋 Standardized field protocols democratize access, allowing landowners and developers to conduct ecosystem monitoring with shipped kits and basic training [4]
- ⚖️ Legal acceptance of eDNA as probable cause is expanding, though regulatory implementation varies significantly by jurisdiction and application [4]
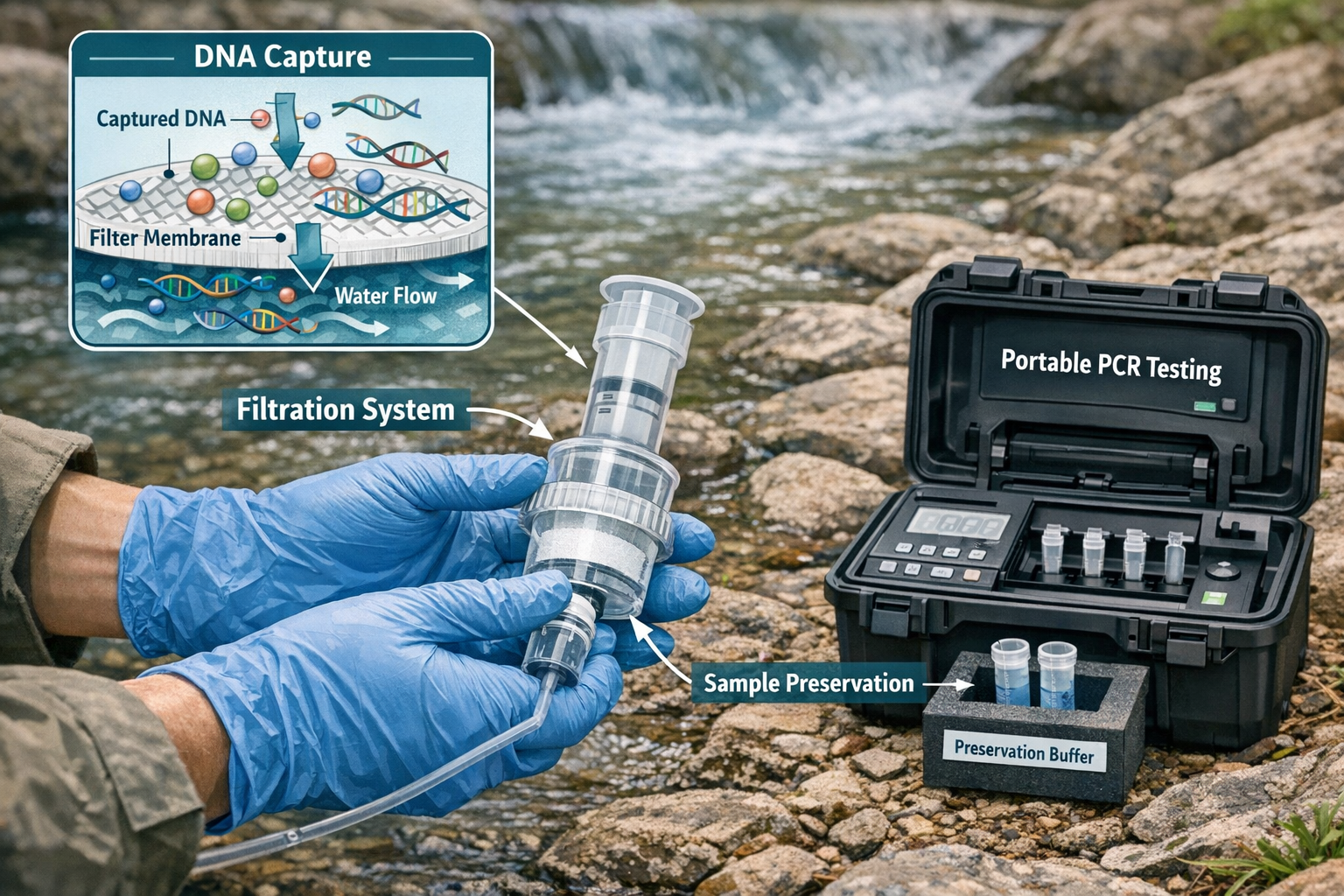
Understanding Molecular Manipulation Impacts in Modern Ecosystems
What Are Molecular Manipulation Impacts?
Molecular manipulation impacts refer to the genetic and ecological changes resulting from deliberate or accidental alterations to organism DNA within ecosystems. These impacts manifest through multiple pathways:
- Gene drive technologies designed to control invasive species populations
- Climate-adapted organisms introduced to strengthen ecosystem resilience
- Genetically modified organisms (GMOs) escaping agricultural systems
- Horizontal gene transfer between modified and wild populations
- Epigenetic changes triggered by environmental stressors
The challenge for biodiversity professionals conducting biodiversity impact assessments lies in detecting these changes early, accurately, and cost-effectively across diverse habitat types.
Why 2026 Marks a Critical Threshold
Several converging factors make 2026 a watershed year for molecular monitoring:
Regulatory pressure has intensified. Environmental agencies increasingly require genetic baseline data before approving development projects. Developers working with biodiversity surveyors must now demonstrate that proposed activities won't disrupt genetic diversity or enable invasive species establishment.
Detection technology has matured. Over 15 years of advancement in molecular methods have expanded eDNA from single-species detection to community-wide biodiversity monitoring, often outperforming traditional practices [1]. What once required specialized university laboratories can now be conducted by trained landowners using standardized field protocols [4].
Legal frameworks are catching up. By 2026, regulatory acceptance of eDNA data for environmental decision-making has grown substantially, though implementation remains uneven across jurisdictions [4]. Some agencies now accept eDNA evidence as probable cause for regulatory action, while others require corroborating traditional survey data.
eDNA Survey Techniques for Detecting Genetic Changes in 2026 Ecosystems
Core eDNA Collection Methodologies
Water sampling remains the most common approach for aquatic and semi-aquatic ecosystems. Standardized protocols now guide collection volume, filtration methods, and preservation techniques:
| Ecosystem Type | Sample Volume | Filter Size | Replicates | Preservation |
|---|---|---|---|---|
| Rivers/streams | 1-2 liters | 0.45-1.5 μm | 3-5 per site | -20°C or ethanol |
| Ponds/lakes | 2-5 liters | 0.45-1.5 μm | 5-8 per site | -20°C or ethanol |
| Marine/coastal | 5-10 liters | 0.45-1.5 μm | 8-12 per site | -80°C preferred |
| Wetlands | 1-3 liters | 1.5 μm | 4-6 per site | -20°C or ethanol |
Soil sampling has expanded rapidly for terrestrial applications. Composite samples from multiple depths (0-5cm, 5-15cm) capture DNA from organisms with different habitat preferences. This approach proves particularly valuable for landowners assessing biodiversity baselines before habitat enhancement projects.
Air sampling represents the frontier of eDNA technology. Specialized filters capture airborne DNA from pollen, spores, skin cells, and respiratory droplets, enabling detection of terrestrial vertebrates and plant communities without physical contact.
Advanced Laboratory Analysis Techniques
Digital droplet PCR (ddPCR) has become the gold standard for 2026 applications requiring high sensitivity. Unlike traditional quantitative PCR (qPCR), ddPCR partitions each sample into thousands of individual droplets, analyzing each separately. This provides superior sensitivity and precision for detecting low-abundance target DNA in complex environmental samples [3].
"The shift from qPCR to ddPCR represents a 10-100x improvement in detection limits, enabling surveyors to identify invasive species before populations establish reproductive footholds." – Environmental Genomics Research Consortium
Metabarcoding workflows sequence multiple species simultaneously from single samples. By targeting conserved gene regions (COI for invertebrates, 16S for bacteria, ITS for fungi, 12S for vertebrates), laboratories generate comprehensive community profiles revealing:
- Species richness (total number of species present)
- Relative abundance (proportional representation of each species)
- Genetic diversity (within-species variation indicating population health)
- Novel sequences (potentially indicating genetic manipulation or hybridization)
Synthetic DNA tracers represent a breakthrough innovation from Cornell researchers who released tracers in Cayuga Lake, tracked movement for 33 hours, and built predictive models showing where sampled eDNA particles originated [1]. Using only 1 microgram of DNA encapsulated in biodegradable polymers, these tracers enable large-scale field studies with minimal environmental impact [1].
Field Implementation Protocols for Molecular Manipulation Impacts

Pre-sampling site assessment determines optimal collection locations. For developers planning construction projects, strategic sampling at upstream, on-site, and downstream locations establishes whether genetic changes originate from the development footprint or external sources.
Contamination prevention protocols have become increasingly rigorous:
- Dedicated equipment for each site prevents cross-contamination
- Bleach and UV sterilization between samples eliminates residual DNA
- Negative controls (filtered sterile water) verify protocol integrity
- Positive controls (known DNA concentrations) confirm detection sensitivity
- Field blanks (open collection bottles) identify airborne contamination
Sample timing considerations account for DNA persistence windows. In most rivers, ponds, and farm reservoirs, environmental DNA degrades within 24-48 hours [4]. Temperature, UV exposure, flow rate, and microbial activity influence degradation rates, requiring surveyors to adjust sampling frequency based on local conditions.
Chain of custody documentation has become essential as eDNA evidence gains legal standing. Detailed records tracking sample collection time, GPS coordinates, environmental conditions, handler identity, and storage temperatures protect the evidentiary value of molecular data.
Interpreting Results for Genetic Change Detection
Baseline comparison analysis forms the foundation of change detection. Surveyors conducting biodiversity net gain assessments establish pre-development genetic profiles, then compare post-construction samples to quantify impacts.
Threshold determination varies by application:
- Invasive species detection: Single positive detection may trigger management response
- Endangered species monitoring: Multiple positive detections confirm presence
- Population health assessment: Genetic diversity metrics compared to reference populations
- GMO escape detection: Novel sequences matched against known modified organisms
False positive and false negative considerations require careful interpretation. Environmental contamination, primer bias, PCR inhibition, and DNA degradation all influence results. Professional surveyors integrate eDNA data with traditional observations and habitat assessments to build robust conclusions.
Molecular Manipulation Impacts: eDNA Applications for 2026 Biodiversity Projects
Invasive Species Early Detection and Management
USGS point-of-use field assays represent a major advancement for invasive species control. Collaborative research involving four USGS science centers has created real-time field detection tests for species like brown treesnakes, requiring minimal equipment and enabling rapid on-site analysis [7].
Post-removal verification has become standard practice. Agencies increasingly deploy eDNA to confirm whether invasive species have been successfully eradicated or whether reinvasion has occurred following control efforts [4]. Most agencies use eDNA to guide follow-up monitoring rather than as standalone confirmation, combining molecular evidence with traditional survey methods.
Cargo and ballast water screening prevents new introductions. Ports now routinely test ballast water discharge for invasive species DNA, rejecting vessels carrying genetic signatures of prohibited organisms [1].
Rare and Endangered Species Monitoring
High sensitivity for low-density populations makes eDNA ideal for endangered species work. Traditional survey methods often fail to detect rare species even when present, leading to false absence conclusions that undermine conservation planning. eDNA technology demonstrates enhanced effectiveness for identifying species at very low densities across aquatic, terrestrial, and atmospheric ecosystems [3].
Non-invasive monitoring reduces disturbance to sensitive species. Rather than capturing, handling, or visually observing endangered animals—activities that may cause stress or habitat disruption—surveyors collect water, soil, or air samples from unoccupied locations.
Population connectivity assessment reveals genetic exchange between habitat patches. By analyzing genetic markers in eDNA samples from multiple sites, surveyors map gene flow patterns, identify isolated populations requiring translocation support, and prioritize habitat corridors for protection.
Commercial Fisheries and Aquaculture Management
Stock assessment applications have expanded rapidly. Environmental managers now use eDNA data to evaluate commercially exploited fish populations, tracking spawning migrations, quantifying recruitment success, and detecting illegal fishing activity [1].
Disease surveillance protects aquaculture operations. Regular eDNA screening detects pathogen presence before clinical disease emerges, enabling early intervention that prevents catastrophic losses.
Escapement monitoring tracks farmed fish entering wild populations. Genetic signatures distinguish hatchery-origin from wild-origin individuals, quantifying the extent of genetic pollution from aquaculture facilities.
Offshore Energy and Infrastructure Impact Assessment
Baseline characterization before construction establishes pre-impact genetic diversity. Developers planning offshore wind farms, tidal energy installations, or subsea cables conduct comprehensive eDNA surveys to document existing communities and identify sensitive species [1].
Construction monitoring detects displacement or mortality events. Increased DNA concentrations during pile driving or dredging indicate organism stress, triggering adaptive management responses.
Long-term compliance verification demonstrates successful mitigation. Post-construction eDNA monitoring confirms that predicted impacts materialized as modeled and that compensation measures achieved intended biodiversity outcomes—critical evidence for achieving biodiversity net gain without risk.
Climate Change Adaptation Monitoring
Range shift detection tracks species moving in response to changing conditions. Regular eDNA sampling along elevation or latitude gradients reveals leading edges of range expansions and trailing edges of contractions.
Assisted migration verification confirms that translocated climate-adapted organisms establish successfully. Following intentional introductions of southern genotypes to northern habitats, eDNA monitoring tracks population establishment without requiring capture-based surveys.
Phenological change assessment detects shifts in breeding, migration, and hibernation timing. Temporal eDNA sampling reveals whether species presence patterns align with historical norms or show climate-driven alterations.
Implementing eDNA Survey Techniques in Biodiversity Net Gain Projects
Integration with Traditional Survey Methods
Complementary rather than replacement approaches yield the most robust results. While eDNA excels at detecting presence, traditional methods provide essential context about abundance, behavior, and habitat use. Surveyors working on biodiversity net gain projects typically combine:
- eDNA screening to identify which species occur on site
- Visual surveys to quantify population sizes and age structures
- Habitat assessment to evaluate quality and carrying capacity
- Acoustic monitoring to document temporal activity patterns
Cost-Benefit Analysis for Developers
Initial investment requirements have decreased substantially. What once cost £5,000-10,000 per site now ranges from £500-2,000 depending on target species and analysis depth. Developers planning small projects find eDNA particularly cost-effective compared to multi-season traditional surveys.
Time savings prove substantial. Traditional bat surveys require multiple visits across breeding seasons. Soil eDNA analysis from a single visit can detect bat presence through guano DNA, reducing survey timelines from 6+ months to 4-6 weeks.
Regulatory acceptance varies. Some planning authorities fully accept eDNA evidence for presence/absence determinations, while others require traditional confirmation. Developers should consult with biodiversity surveyors familiar with local requirements before committing to eDNA-only approaches.
Landowner Applications for Habitat Banking
Baseline documentation establishes starting conditions for landowners creating biodiversity units. Comprehensive eDNA surveys generate genetic inventories demonstrating habitat value and enhancement potential.
Enhancement verification proves that management interventions achieve intended outcomes. Following wetland restoration or woodland creation, eDNA monitoring tracks colonization by target species, providing evidence that supports biodiversity unit sales.
Long-term monitoring efficiency reduces ongoing costs. Rather than hiring specialists for annual traditional surveys, landowners can collect eDNA samples using standardized protocols and ship to laboratories for analysis—democratizing access to professional-grade monitoring [4].
Quality Assurance and Accreditation Standards
ISO-compliant laboratory selection ensures reliable results. Laboratories processing eDNA samples should maintain:
- ISO 17025 accreditation for testing and calibration
- Documented quality management systems with internal audits
- Proficiency testing participation through inter-laboratory comparisons
- Positive and negative control protocols for every analytical run
- Data management systems with full traceability and archiving
Field technician certification programs have emerged across the UK. Organizations like the Chartered Institute of Ecology and Environmental Management (CIEEM) now offer eDNA-specific training modules covering collection protocols, contamination prevention, and chain of custody procedures.
Data reporting standards facilitate regulatory review. Comprehensive eDNA reports should include sampling locations (GPS coordinates), environmental conditions (temperature, pH, conductivity), collection methods, laboratory protocols, quality control results, taxonomic identifications, and interpretation limitations.
Future Directions for Molecular Manipulation Impacts and eDNA Survey Techniques
Emerging Technologies on the Horizon
Portable sequencing devices will enable real-time field analysis. Oxford Nanopore MinION sequencers already allow on-site DNA sequencing, with ongoing development aimed at reducing cost, improving accuracy, and expanding reference databases.
Artificial intelligence integration will automate species identification. Machine learning algorithms trained on genetic sequences can rapidly match environmental samples against comprehensive databases, flagging novel sequences that may indicate genetic manipulation or undescribed species.
Quantitative abundance estimation remains challenging but improving. Current eDNA methods excel at presence/absence determination but struggle with accurate abundance quantification due to variable DNA shedding rates, degradation, and transport. Ongoing research combining synthetic tracers with predictive models aims to establish reliable abundance conversion factors [1].
Regulatory Evolution and Standardization
International standardization efforts are accelerating. The International Organization for Standardization (ISO) has published technical specifications for eDNA sampling and analysis, with full standards expected by 2027-2028.
Legal precedent establishment continues unevenly. Court cases testing eDNA evidence admissibility are creating jurisdiction-specific precedents that will shape future regulatory frameworks. Surveyors should monitor developments in their operating regions.
Integration with biodiversity net gain metrics requires ongoing refinement. Current biodiversity metric calculations emphasize habitat condition and distinctiveness but don't explicitly account for genetic diversity. Future iterations may incorporate molecular data to reward sites with high genetic variation.
Professional Development Opportunities
Specialized training programs are expanding rapidly. Universities, professional organizations, and commercial providers now offer:
- Certificate courses in eDNA sampling and analysis (1-5 days)
- Master's programs in conservation genomics (1-2 years)
- Online modules covering specific taxonomic groups or applications
- Field workshops providing hands-on experience with collection protocols
Career pathways are diversifying. Demand is growing for professionals who bridge traditional ecological survey skills with molecular expertise—creating opportunities for established surveyors to expand service offerings and for early-career professionals to differentiate themselves in competitive markets.
Conclusion: Embracing Molecular Tools for 2026 Conservation Challenges
The Molecular Manipulation Impacts: eDNA Survey Techniques for Genetic Changes in 2026 Ecosystems represent more than technological advancement—they embody a fundamental shift in how biodiversity professionals understand, monitor, and protect natural systems. As molecular manipulation technologies increasingly reshape ecosystems, the ability to detect and track genetic changes has evolved from specialized research capability to essential professional competency.
For developers navigating biodiversity net gain requirements, eDNA survey techniques offer faster, more cost-effective, and often more sensitive alternatives to traditional methods. The technology's maturation—from Cornell's predictive models tracking DNA movement [1] to USGS field assays enabling real-time detection [7]—has created practical tools ready for immediate implementation.
For landowners managing habitat banks or pursuing sustainable land use practices, eDNA monitoring provides accessible, affordable means to document biodiversity value and verify enhancement outcomes. The democratization of molecular tools through standardized protocols and shipped testing kits [4] removes barriers that previously limited ecosystem monitoring to well-funded organizations.
For conservation professionals, the integration of eDNA techniques with traditional survey methods creates comprehensive assessment frameworks capable of detecting both ecological and genetic changes. This holistic approach positions surveyors to address 2026's most pressing challenges—from invasive species management to climate change adaptation.
Actionable Next Steps
For developers and planners:
- Consult with biodiversity surveyors about incorporating eDNA into baseline assessments
- Request cost-benefit analyses comparing eDNA to traditional survey approaches
- Verify local planning authority acceptance of molecular evidence
For landowners:
- Establish genetic baselines before implementing habitat management
- Explore eDNA monitoring for biodiversity unit verification
- Consider citizen science programs that provide training and equipment
For conservation professionals:
- Pursue specialized training in eDNA collection and interpretation
- Develop partnerships with accredited molecular laboratories
- Stay current with evolving regulatory frameworks and standardization efforts
The molecular revolution in conservation science has arrived. Those who embrace these tools position themselves at the forefront of 2026's biodiversity challenges, equipped to detect changes invisible to traditional methods and to verify outcomes with unprecedented precision. The question is no longer whether to adopt eDNA survey techniques, but how quickly professionals can integrate them into standard practice.
References
[1] Environmental Dna Breakthrough Will Aid Conservation Efforts – https://news.cornell.edu/stories/2026/01/environmental-dna-breakthrough-will-aid-conservation-efforts
[3] Pmc12789655 – https://pmc.ncbi.nlm.nih.gov/articles/PMC12789655/
[4] Edna Testing For Invasive Species – https://landwildlifereport.com/2026/01/20/edna-testing-for-invasive-species/


