Below the visible surface of every grassland lies an invisible ecosystem worth billions—mycorrhizal fungi networks that connect plant roots across entire meadows, transferring nutrients and information through microscopic highways. Yet current Biodiversity Net Gain (BNG) assessments routinely overlook these underground architects, potentially undermining the accuracy of baseline calculations and the long-term success of habitat restoration projects. As 2026 brings heightened scrutiny to biodiversity assessment methodologies, understanding Grassland Fungi Networks in Biodiversity Net Gain Baselines: Underground Survey Techniques for 2026 has become essential for developers, ecologists, and landowners committed to genuine ecological enhancement.
The integration of fungal network mapping into BNG protocols represents a paradigm shift in how professionals evaluate grassland quality. Traditional above-ground surveys capture only half the story—the visible plants, invertebrates, and birds. Meanwhile, the mycorrhizal networks that determine soil health, carbon sequestration capacity, and ecosystem resilience remain hidden from standard assessment frameworks. This article reveals the cutting-edge methods for mapping these underground networks using soil cores and DNA metabarcoding, providing practical guidance for incorporating fungal baseline data into comprehensive BNG reports.
Key Takeaways
- 🍄 Mycorrhizal networks form the foundation of grassland soil health but are absent from most BNG baseline assessments
- 🔬 Soil core sampling combined with DNA metabarcoding provides accurate fungal diversity and abundance data for baseline calculations
- 📊 Fungal metrics can enhance BNG unit calculations by revealing true habitat condition and restoration potential
- 🌱 Underground biodiversity directly influences above-ground plant diversity and ecosystem services valued in BNG frameworks
- ⚖️ Regulatory frameworks in 2026 increasingly recognize soil biodiversity as critical to achieving 10% biodiversity net gain targets

Understanding Mycorrhizal Networks in Grassland Ecosystems
Mycorrhizal fungi form symbiotic relationships with approximately 90% of plant species, including most grassland flora. These fungi extend plant root systems through thread-like structures called hyphae, creating vast underground networks that can span several hectares. In return for carbon compounds from plants, mycorrhizal fungi provide essential services:
Key Functions of Grassland Fungal Networks:
- Nutrient acquisition: Fungi access phosphorus, nitrogen, and micronutrients beyond root reach
- Water transport: Hyphal networks improve drought resistance by extending water absorption zones
- Carbon storage: Fungal biomass sequesters significant carbon in stable soil compounds
- Plant communication: Networks facilitate resource sharing and stress signal transmission between plants
- Soil structure: Fungal hyphae bind soil particles, reducing erosion and improving water infiltration
Why Fungal Networks Matter for BNG Baselines
The presence, diversity, and abundance of mycorrhizal fungi directly indicate grassland habitat quality. Species-rich grasslands with intact fungal communities demonstrate:
✅ Higher resilience to environmental stressors
✅ Greater carbon sequestration potential
✅ Enhanced nutrient cycling efficiency
✅ Improved water regulation services
✅ Superior restoration outcomes when used as receptor sites
When conducting biodiversity impact assessments, excluding fungal network data means missing critical indicators of ecosystem function. A grassland may appear floristically diverse above ground while harboring degraded fungal communities that compromise long-term habitat viability. This disconnect can lead to overestimated baseline values and underperforming restoration schemes.
According to recent policy developments, regulators increasingly recognize soil biodiversity as integral to BNG compliance [1]. Natural England guidance now encourages assessments that extend beyond traditional habitat surveys to incorporate soil health metrics, creating opportunities for fungal network data integration.
Soil Core Sampling Protocols for Grassland Fungi Networks in Biodiversity Net Gain Baselines
Accurate fungal baseline assessment begins with proper soil sampling. Unlike above-ground surveys that rely on visual observation, underground biodiversity requires physical sample collection and laboratory analysis. The following protocols represent current best practices for Grassland Fungi Networks in Biodiversity Net Gain Baselines: Underground Survey Techniques for 2026.
Sampling Design and Site Selection
Stratified Random Sampling Approach:
- Divide the survey area into representative habitat zones based on vegetation composition, soil type, and management history
- Establish sampling points using a grid system with minimum 20-meter spacing
- Collect 5-10 cores per hectare for statistically robust baseline data
- Document GPS coordinates and environmental variables at each sampling location
| Grassland Type | Recommended Cores/Hectare | Sampling Depth | Optimal Season |
|---|---|---|---|
| Species-rich meadow | 8-10 | 0-15 cm | Late spring/early autumn |
| Semi-improved grassland | 6-8 | 0-15 cm | Spring or autumn |
| Lowland calcareous grassland | 10-12 | 0-20 cm | Late spring |
| Acid grassland | 6-8 | 0-15 cm | Autumn |
Field Collection Techniques
Equipment Required:
- Stainless steel soil corers (diameter 2-5 cm)
- Sterile sample bags or tubes
- GPS device with sub-meter accuracy
- Field datasheets and waterproof markers
- Portable cooler with ice packs
- Digital camera for site documentation
Step-by-Step Collection Process:
- Clear surface vegetation without disturbing soil structure
- Insert corer vertically to target depth using steady downward pressure
- Extract core carefully to maintain soil horizon integrity
- Transfer immediately to sterile container, avoiding contamination
- Label comprehensively with site code, coordinates, date, and depth
- Store at 4°C and transport to laboratory within 24 hours
- Record environmental data: soil moisture, temperature, pH, vegetation cover
Expert Tip: Collect cores when soil moisture is moderate—neither saturated nor excessively dry. Extreme conditions compromise fungal DNA preservation and extraction efficiency.
Sample Processing and Storage
Upon laboratory arrival, samples require careful processing to preserve fungal DNA integrity:
- Homogenize each core by removing stones and large organic debris
- Subsample 10-20 grams for DNA extraction
- Store subsamples at -20°C for molecular analysis
- Archive remaining soil at 4°C for potential physicochemical analysis
- Document chain of custody for quality assurance
When planning BNG projects, budget adequate time for sample collection during appropriate seasonal windows. Fungal communities exhibit temporal variation, so baseline surveys should ideally capture peak growing season diversity.

DNA Metabarcoding and Analysis Methods for Underground Survey Techniques
DNA metabarcoding has revolutionized fungal biodiversity assessment by enabling comprehensive community characterization from soil samples. This molecular approach identifies hundreds of fungal species simultaneously, providing unprecedented detail for Grassland Fungi Networks in Biodiversity Net Gain Baselines: Underground Survey Techniques for 2026.
DNA Extraction and Amplification
Laboratory Workflow:
- DNA Extraction: Use commercial soil DNA extraction kits optimized for high humic acid content
- Quality Assessment: Verify DNA concentration and purity using spectrophotometry
- PCR Amplification: Target fungal ITS (Internal Transcribed Spacer) regions using universal primers
- Library Preparation: Attach unique molecular identifiers to each sample for multiplexing
- Sequencing: Submit to high-throughput sequencing platform (Illumina MiSeq or NovaSeq)
Key Molecular Markers for Fungal Identification:
- ITS1 and ITS2 regions: Primary barcode for fungal species identification
- 18S rRNA gene: Provides phylogenetic context for broader taxonomic groups
- LSU (Large Subunit) rRNA: Useful for arbuscular mycorrhizal fungi characterization
Bioinformatics Pipeline
Raw sequence data requires sophisticated computational analysis to generate meaningful ecological metrics:
Data Processing Steps:
- Quality filtering: Remove low-quality sequences and chimeras
- Clustering: Group similar sequences into Operational Taxonomic Units (OTUs) or Amplicon Sequence Variants (ASVs)
- Taxonomic assignment: Compare sequences against reference databases (UNITE, GenBank)
- Diversity calculation: Generate alpha and beta diversity metrics
- Functional annotation: Assign ecological guilds (mycorrhizal, saprotrophic, pathogenic)
Interpreting Fungal Diversity Metrics for BNG
Alpha Diversity Indicators (within-sample diversity):
- Species richness: Total number of fungal taxa detected
- Shannon diversity index: Accounts for both richness and evenness
- Simpson's index: Emphasizes dominant species presence
Beta Diversity Indicators (between-sample variation):
- Bray-Curtis dissimilarity: Compares community composition across sites
- UniFrac distances: Incorporates phylogenetic relationships between taxa
Functional Diversity Metrics:
- Mycorrhizal colonization potential: Proportion of mycorrhizal fungi in community
- Saprotrophic capacity: Abundance of decomposer fungi indicating nutrient cycling
- Rare species richness: Presence of specialist fungi indicating habitat quality
Establishing Baseline Thresholds
To integrate fungal data into BNG assessment frameworks, establish condition-based thresholds:
| Habitat Condition | Mycorrhizal Taxa Richness | Shannon Diversity | Rare Species (%) |
|---|---|---|---|
| Good | >80 taxa | >3.5 | >15% |
| Moderate | 50-80 taxa | 2.5-3.5 | 8-15% |
| Poor | <50 taxa | <2.5 | <8% |
These thresholds should be calibrated against regional reference grasslands and adjusted for grassland type. Species-rich calcareous grasslands naturally support higher fungal diversity than improved agricultural grasslands.
Practical Application:
When baseline surveys reveal degraded fungal communities despite reasonable above-ground vegetation, this indicates:
⚠️ Historical management impacts (fertilization, herbicide use) affecting soil biology
⚠️ Limited restoration potential without soil remediation interventions
⚠️ Reduced ecosystem service delivery compared to visually similar sites
⚠️ Lower appropriate baseline values for BNG unit calculations
This information enables more accurate biodiversity unit valuations and realistic enhancement targets.
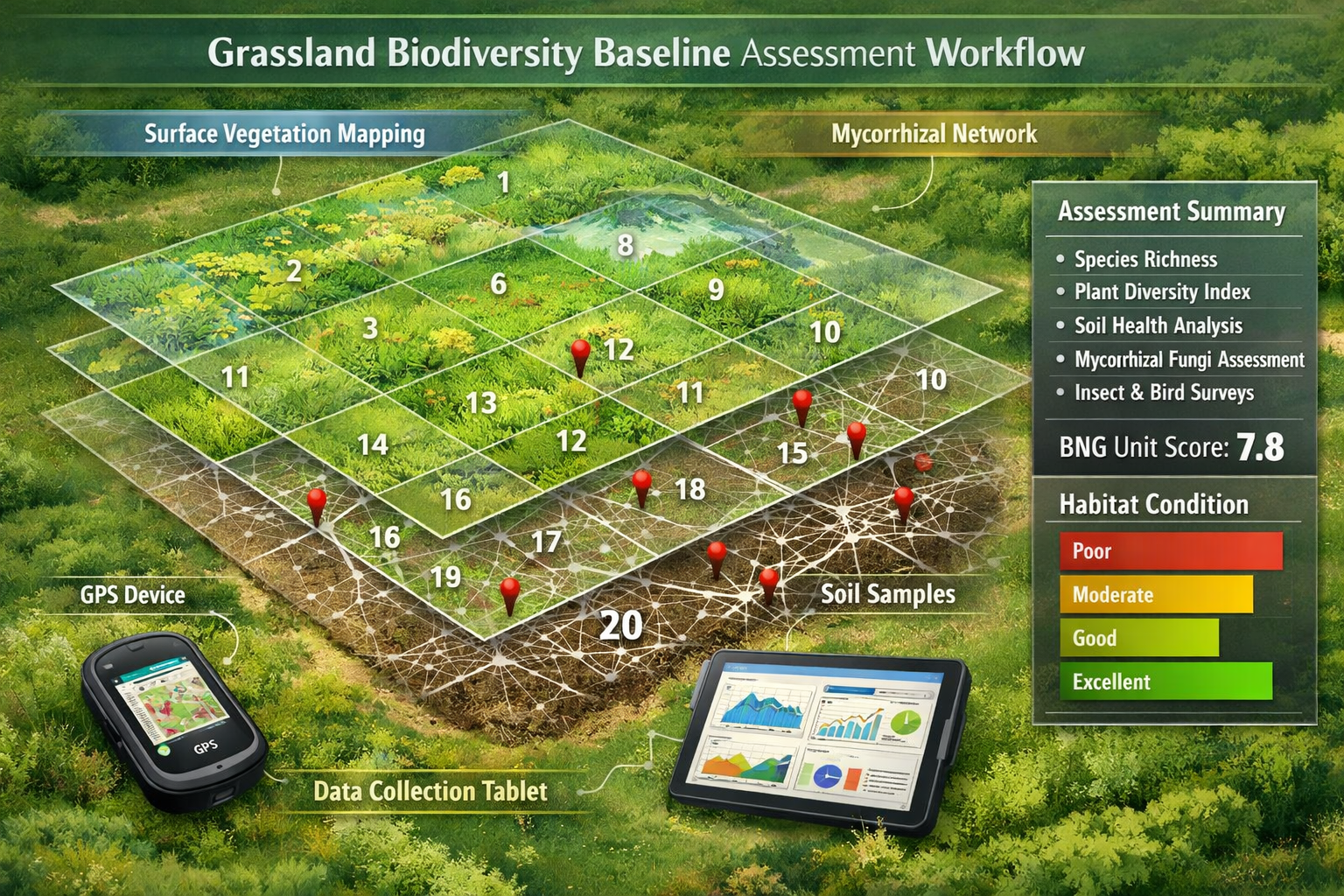
Integrating Fungal Network Data into BNG Assessment Frameworks
The challenge facing practitioners in 2026 is translating fungal diversity metrics into standardized BNG calculations. While current statutory biodiversity metric frameworks primarily focus on habitat type and condition, emerging best practices recognize soil biodiversity as a supplementary indicator.
Modifying Condition Assessment Criteria
Enhanced Condition Scoring:
Traditional grassland condition assessments evaluate:
- Plant species richness and composition
- Sward structure and height variation
- Physical damage and disturbance indicators
- Management appropriateness
Supplementary Fungal Indicators:
Add soil biodiversity criteria to condition assessment sheets:
✓ Mycorrhizal diversity score (based on taxa richness thresholds)
✓ Functional group representation (presence of arbuscular, ectomycorrhizal, and ericoid types)
✓ Rare species indicators (specialist fungi associated with high-quality grasslands)
✓ Pathogen-to-beneficial ratio (low pathogen abundance indicates healthy systems)
These supplementary criteria can adjust habitat condition scores within existing frameworks, typically influencing the final condition category by one level (e.g., upgrading "moderate" to "good" condition when fungal metrics exceed thresholds).
Documentation Requirements for BNG Reports
When incorporating fungal baseline data into BNG reports, include:
Essential Documentation:
- Sampling methodology: Detailed protocol including core numbers, depths, and spatial distribution
- Laboratory procedures: DNA extraction methods, sequencing platform, and bioinformatics pipeline
- Reference standards: Comparison with regional benchmark grasslands or published datasets
- Quality assurance: Positive and negative controls, replication strategy, statistical power analysis
- Raw data availability: Sequence data deposited in public repositories (NCBI, EBI)
- Interpretation framework: Clear explanation of how fungal metrics influenced condition assessment
Regulatory Considerations in 2026
Current BNG regulations do not mandate fungal network assessment, but policy evolution suggests growing recognition of soil biodiversity importance [1]. Forward-thinking developers and landowners incorporating fungal baselines gain several advantages:
Strategic Benefits:
- 🎯 Stronger evidence base for habitat condition claims
- 🎯 Differentiation in competitive markets for biodiversity unit sales
- 🎯 Reduced risk of baseline overestimation and subsequent compliance issues
- 🎯 Enhanced credibility with regulators and stakeholders
- 🎯 Future-proofing against evolving assessment standards
Organizations like Plantlife have advocated for soil biodiversity integration in BNG frameworks, particularly for grassland habitats where fungal networks critically influence ecosystem function [4]. As assessment methodologies mature, early adoption of comprehensive underground surveys positions projects favorably for regulatory approval.
Practical Challenges and Solutions for Underground Survey Techniques
Implementing Grassland Fungi Networks in Biodiversity Net Gain Baselines: Underground Survey Techniques for 2026 presents several practical challenges that require strategic solutions.
Cost Considerations
Budget Components:
- Field sampling: £500-£1,500 per site (depending on area and core numbers)
- Laboratory analysis: £150-£300 per sample for DNA extraction and sequencing
- Bioinformatics: £1,000-£3,000 for computational analysis and reporting
- Total per hectare: £2,000-£5,000 for comprehensive fungal baseline assessment
Cost Management Strategies:
💡 Composite sampling: Pool cores from similar habitat zones to reduce analysis costs
💡 Phased implementation: Prioritize high-value or sensitive sites for detailed fungal assessment
💡 Collaborative approaches: Share sequencing runs with other projects to reduce per-sample costs
💡 Strategic targeting: Focus fungal surveys on grasslands where baseline uncertainty is highest
When planning development projects, allocate 5-10% of total ecological survey budgets to soil biodiversity assessment for grassland-dominated sites.
Technical Expertise Requirements
Fungal network assessment requires specialized expertise spanning:
- Field ecology: Understanding grassland habitats and sampling design
- Molecular biology: DNA extraction, PCR, and sequencing technologies
- Bioinformatics: Computational analysis of high-throughput sequencing data
- Mycology: Fungal taxonomy and ecological interpretation
Solutions:
🔧 Partner with specialist consultancies offering integrated soil biodiversity services
🔧 Collaborate with academic institutions conducting fungal ecology research
🔧 Commission commercial laboratories providing turnkey metabarcoding services
🔧 Invest in training for in-house ecology teams on soil sampling protocols
Temporal Variability Management
Fungal communities exhibit seasonal and annual variation influenced by:
- Temperature and moisture regimes
- Plant phenology and root activity
- Management interventions (grazing, cutting)
- Weather patterns and climate fluctuation
Standardization Approaches:
📅 Consistent seasonal timing: Conduct baseline and post-intervention surveys during comparable periods
📅 Multi-season sampling: Capture spring and autumn communities for comprehensive baseline
📅 Long-term monitoring: Establish permanent sampling points for temporal tracking
📅 Reference site comparison: Use contemporaneous samples from benchmark grasslands
Data Interpretation Uncertainties
Fungal taxonomy and ecology remain active research areas, creating interpretation challenges:
- Incomplete reference databases for some fungal groups
- Functional guild assignments based on limited ecological data
- Regional variation in fungal community composition
- Complex interactions between fungi, plants, and soil conditions
Mitigation Strategies:
🔍 Conservative interpretation: Focus on well-characterized taxa and robust diversity metrics
🔍 Expert consultation: Engage mycologists for specialized taxonomic and ecological guidance
🔍 Comparative approaches: Emphasize relative differences between sites rather than absolute values
🔍 Continuous learning: Update interpretation frameworks as fungal ecology knowledge advances
Future Directions for Fungal Network Assessment in BNG
The integration of underground biodiversity into BNG frameworks represents an evolving frontier. Several developments will shape Grassland Fungi Networks in Biodiversity Net Gain Baselines: Underground Survey Techniques for 2026 and beyond:
Technological Advances
Emerging Methods:
- Environmental DNA (eDNA) approaches: Non-invasive sampling from soil water extracts
- Portable sequencing devices: Field-deployable DNA analysis using Oxford Nanopore technology
- Hyperspectral imaging: Remote sensing of soil fungal activity through vegetation spectral signatures
- Artificial intelligence: Machine learning models predicting fungal diversity from environmental variables
Standardization Initiatives
Industry bodies and research institutions are developing:
- Standardized sampling protocols for soil biodiversity in BNG contexts
- Reference databases of fungal communities from UK grassland types
- Condition assessment frameworks incorporating soil biological metrics
- Training programs for ecological consultants on underground survey techniques
Policy Evolution
Regulatory frameworks increasingly recognize soil health as fundamental to biodiversity outcomes. The 2026 policy landscape shows movement toward:
- Mandatory soil biodiversity assessment for certain habitat types
- Soil carbon and fungal network metrics in ecosystem service valuations
- Enhanced biodiversity unit values for sites with documented healthy soil communities
- Off-site delivery requirements specifying soil quality standards
Organizations seeking to achieve biodiversity net gain without risk should monitor these policy developments and consider proactive adoption of comprehensive baseline assessment approaches.
Research Priorities
Key knowledge gaps requiring investigation:
- Quantitative relationships between fungal diversity and ecosystem service delivery
- Restoration trajectories for degraded fungal communities in different grassland types
- Cost-benefit analysis of fungal network enhancement interventions
- Fungal indicator species for rapid habitat quality assessment
- Integration of fungal metrics with existing biodiversity metric calculations
Conclusion
Grassland Fungi Networks in Biodiversity Net Gain Baselines: Underground Survey Techniques for 2026 represent a critical evolution in ecological assessment methodology. The invisible mycorrhizal networks threading through grassland soils provide essential ecosystem functions that determine habitat quality, restoration potential, and long-term biodiversity outcomes. By incorporating soil core sampling and DNA metabarcoding into baseline surveys, practitioners can generate more accurate habitat valuations and design more effective enhancement strategies.
The techniques outlined in this article—from stratified sampling protocols to bioinformatics pipelines—provide practical pathways for integrating fungal diversity data into BNG frameworks. While current regulations do not mandate underground biodiversity assessment, the strategic advantages of comprehensive baseline characterization are substantial: reduced compliance risk, enhanced credibility, and future-proofing against evolving standards.
Actionable Next Steps
For developers, landowners, and ecological consultants working on BNG projects in 2026:
- Evaluate your current baseline assessment protocols to identify gaps in soil biodiversity characterization
- Engage specialist consultancies with molecular ecology expertise for pilot fungal network surveys
- Incorporate soil sampling into field survey schedules during optimal seasonal windows
- Budget appropriately for laboratory analysis and bioinformatics services (£2,000-£5,000 per hectare)
- Document methodologies comprehensively in BNG reports to demonstrate assessment rigor
- Monitor policy developments regarding soil biodiversity requirements in BNG frameworks
- Consider fungal enhancement interventions such as mycorrhizal inoculation or reduced soil disturbance in restoration schemes
The underground world of grassland fungi networks holds the key to truly sustainable biodiversity outcomes. By bringing these hidden ecosystems into the light through rigorous survey techniques, the BNG framework can deliver on its promise of genuine ecological enhancement rather than superficial habitat manipulation. As assessment methodologies mature and regulatory frameworks evolve, those who invest in comprehensive underground surveys today will lead the biodiversity net gain sector tomorrow.
For expert guidance on comprehensive biodiversity net gain assessments that incorporate cutting-edge soil biodiversity techniques, consult with specialists who understand both regulatory requirements and ecological reality.
References
[1] The State Of Biodiversity Net Gain In 2026 Key Policy Updates – https://biodiversity-netgain.co.uk/the-state-of-biodiversity-net-gain-in-2026-key-policy-updates/
[2] Final Report Draft Biodiversity Duty Report 2026 – https://www.solihull.gov.uk/sites/default/files/2026-03/Final-Report-Draft-Biodiversity-Duty-Report-2026.pdf
[3] P1bw615x6ambfzuq – https://docs.planning.org.uk/20260330/146/TB9P7YGM06N00/p1bw615x6ambfzuq.pdf
[4] Biodiversity Net Gain For Landowners – https://www.plantlife.org.uk/our-work/biodiversity-net-gain-for-landowners/


