Chytrid fungus has driven 90 species of amphibians to extinction and threatens hundreds more worldwide—making it the most devastating wildlife disease ever recorded. As Biodiversity Net Gain (BNG) legislation mandates wetland creation and enhancement across England in 2026, ecologists face a critical challenge: establishing baseline amphibian populations without introducing or overlooking this silent killer. Amphibian Chytrid Fungus Surveys in Wetland BNG Projects: Early Detection Strategies for Ecologists represents an essential safeguard that protects both project integrity and native wildlife populations from catastrophic decline.
The stakes extend beyond conservation ethics. Contaminated wetland BNG sites can fail to deliver promised biodiversity units, triggering compliance issues and financial penalties for developers. Early detection through systematic surveillance prevents baseline contamination and ensures that biodiversity net gain assessments reflect genuine ecological value rather than disease-compromised habitats.

Key Takeaways
🔬 Dual-pathogen surveillance: Modern protocols must screen for both Batrachochytrium dendrobatidis (Bd) and Batrachochytrium salamandrivorans (Bsal), with the latter posing emerging threats to UK newt populations
💧 eDNA expansion: Environmental DNA sampling complements traditional swabbing, enabling landscape-scale detection without handling every individual amphibian
📊 Risk mapping integration: Geographic risk models inform survey intensity and biosecurity protocols, concentrating resources on high-vulnerability sites
⚡ Rapid response windows: Early detection during BNG baseline surveys creates intervention opportunities before pathogen establishment threatens project outcomes
🛡️ Biosecurity by design: Prevention protocols must be embedded in BNG project planning from initial site assessments through long-term monitoring phases
Understanding Chytrid Fungus Threats in BNG Wetland Contexts
The Dual Pathogen Challenge
Two distinct chytrid species threaten amphibian populations in wetland BNG projects. Batrachochytrium dendrobatidis (Bd) has established endemic presence across much of the UK, causing chytridiomycosis that disrupts skin function and electrolyte balance in frogs, toads, and newts. The infection compromises the amphibian's ability to regulate water and breathe through their permeable skin, leading to cardiac arrest in severe cases.
The newer threat, Batrachochytrium salamandrivorans (Bsal), targets salamanders and newts specifically. First identified in 2013, this pathogen has devastated fire salamander populations in continental Europe with mortality rates exceeding 90%. While not yet confirmed as established in the UK, its detection would trigger immediate containment responses given the vulnerability of native newt species.
Research programs like the Minnesota Student Network for Amphibian Pathogen Surveillance have tested over 200 amphibian samples for both pathogens, establishing protocols now adaptable to UK wetland contexts[1]. Their surveillance framework demonstrates that systematic monitoring can detect emerging infections before population-level impacts become irreversible.
Why BNG Wetlands Require Heightened Vigilance
Wetland creation and enhancement projects under BNG legislation create ideal conditions for chytrid transmission:
- High amphibian density: Constructed breeding ponds concentrate individuals during spawning seasons
- Stress factors: Newly established habitats may lack optimal conditions, compromising immune function
- Equipment vectors: Survey gear, construction machinery, and materials can transfer infectious zoospores between sites
- Water connectivity: Drainage systems and flood events facilitate pathogen spread across project boundaries
The biodiversity plan development process must incorporate disease surveillance as a standard component, not an afterthought. Baseline surveys that fail to detect existing infections risk attributing future population declines to habitat quality rather than disease, undermining the ecological validity of biodiversity unit calculations.
Amphibian Chytrid Fungus Surveys in Wetland BNG Projects: Swab-Based Detection Protocols
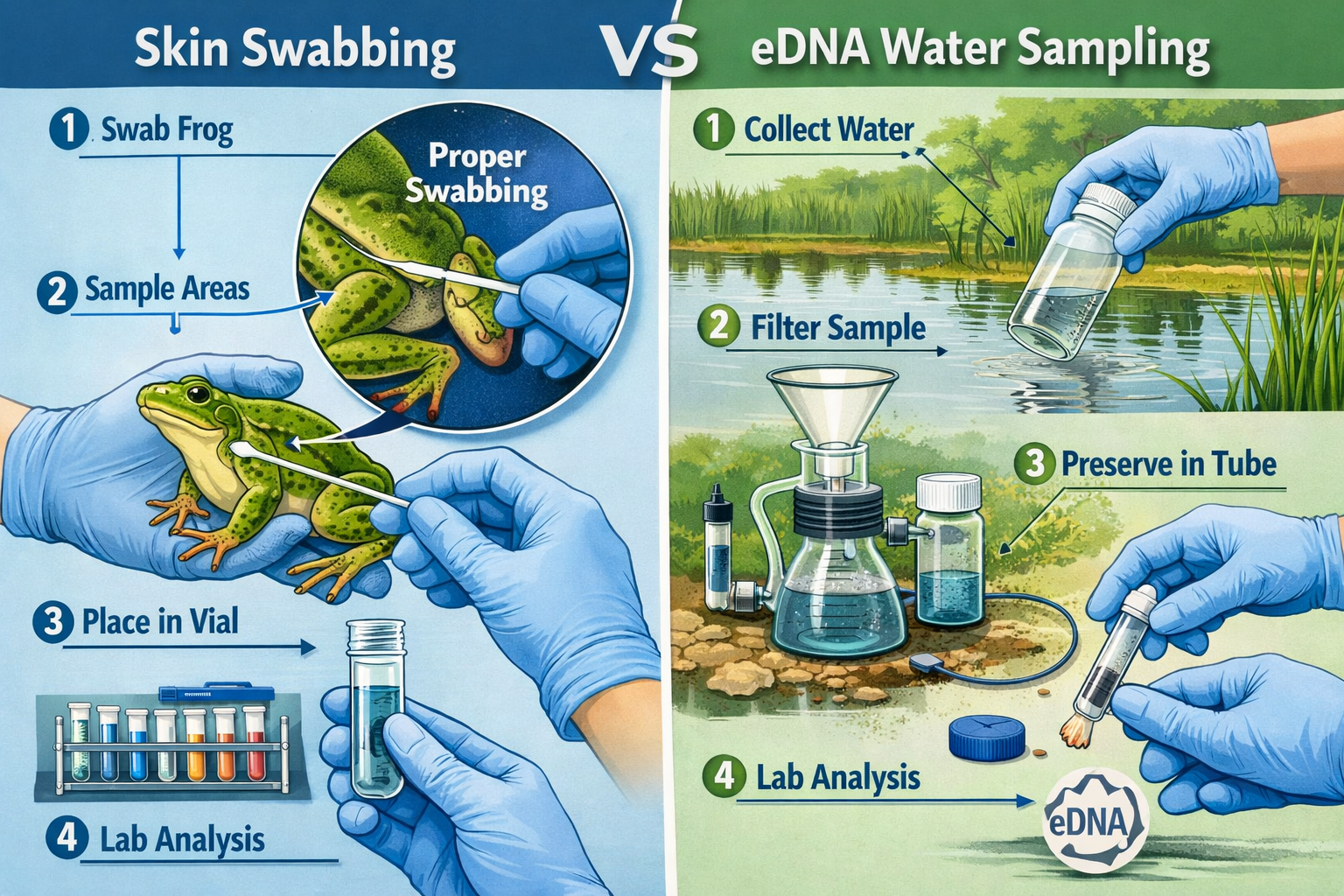
Standard Swabbing Methodology
The gold standard for chytrid detection involves individually swabbing amphibians and analyzing samples through real-time TaqMan PCR (polymerase chain reaction) technology[3]. This molecular approach identifies pathogen DNA with high sensitivity and specificity.
Field Collection Protocol:
- Capture and restraint: Use clean nitrile gloves for each individual to prevent cross-contamination
- Swabbing technique: Stroke sterile cotton swab across ventral surfaces (belly, inner thighs, feet) 5-10 times per area
- Sample preservation: Place swab in sterile tube with preservation buffer or freeze immediately
- Documentation: Record species, life stage, location coordinates, and visual health indicators
- Release: Return amphibian to exact capture location after minimal handling time
Target Sample Sizes:
- Small ponds (<100m²): Minimum 15-20 individuals across species present
- Medium wetlands (100-500m²): 30-40 individuals with proportional species representation
- Large complexes (>500m²): 50+ individuals stratified across habitat zones
The U.S. Geological Survey's national baseline effort (May 2014–August 2017) found no detected cases of B. salamandrivorans across sampled sites, demonstrating that systematic surveillance can establish disease-free status when properly executed[3]. This precedent validates the investment in comprehensive screening for BNG projects.
Seasonal Timing Considerations
Survey timing dramatically affects detection success rates:
Optimal windows:
- Spring (March-May): Peak breeding activity concentrates amphibians at aquatic sites, maximizing capture efficiency
- Autumn (September-October): Juvenile dispersal provides access to current-year recruitment
- Avoid: Mid-summer when many species are terrestrial and winter when amphibians are torpid
Chytrid fungi exhibit temperature-dependent growth, with Bd thriving in cooler conditions (4-25°C) and Bsal preferring slightly warmer ranges (10-20°C). Spring surveys capture peak infection loads when pathogen reproduction rates are highest, improving detection sensitivity.
Laboratory Analysis Standards
Extracted DNA from swabs undergoes real-time TaqMan PCR analysis targeting species-specific genetic markers[3]. This technique amplifies target DNA sequences, allowing detection of even low pathogen loads that might not yet cause clinical disease.
Key laboratory specifications:
- Sensitivity threshold: Detection of ≥1 zoospore equivalent
- Species differentiation: Separate assays for Bd and Bsal
- Quality controls: Positive and negative standards in each run
- Turnaround time: 5-10 business days for standard processing
Results are typically reported as:
- Negative: No pathogen DNA detected
- Positive: Infection confirmed with quantitative load estimate
- Inconclusive: Resampling recommended
The Minnesota surveillance program shares test results through the Amphibian Disease Portal, creating transparency and contributing to regional disease mapping[1]. BNG projects should similarly integrate findings into national monitoring databases to build collective intelligence on pathogen distribution.
Amphibian Chytrid Fungus Surveys in Wetland BNG Projects: eDNA Expansion Methods

Environmental DNA Sampling Advantages
Environmental DNA (eDNA) methods represent a powerful complement to traditional swabbing, particularly for large or complex wetland BNG sites[1]. This approach detects pathogen genetic material shed into water, enabling landscape-scale surveillance without capturing every individual amphibian.
eDNA Benefits for BNG Projects:
✅ Reduced handling stress: Minimizes direct contact with vulnerable populations
✅ Broader spatial coverage: Single water sample integrates signal from multiple individuals
✅ Early warning capability: Detects pathogen presence before clinical disease manifestation
✅ Cost efficiency: Fewer field hours required compared to exhaustive capture efforts
✅ Repeat sampling: Non-invasive nature allows frequent monitoring without population impact
eDNA Collection Protocol
Water Sampling Method:
- Site selection: Collect from areas with concentrated amphibian activity (breeding pools, basking zones)
- Volume requirements: Filter 1-2 liters of water per sample location
- Filtration: Use 0.45μm pore size filters to capture pathogen DNA
- Preservation: Store filters in preservation buffer or freeze immediately
- Replication: Minimum 3 samples per water body to account for spatial heterogeneity
Substrate Sampling:
Sediment and biofilm samples can also yield pathogen DNA, particularly in systems where chytrid zoospores encyst on surfaces. Collect surface sediment (top 2cm) and submerged vegetation samples using sterile technique.
Interpreting eDNA Results
Environmental DNA detection indicates pathogen presence in the environment but doesn't confirm active infections in amphibian populations. Positive eDNA findings should trigger follow-up swabbing to:
- Confirm infections in host populations
- Quantify prevalence rates
- Assess clinical disease manifestation
- Inform intervention decisions
The Minnesota team's planned eDNA surveillance expansion demonstrates growing confidence in this methodology for early detection programs[1]. As techniques mature, eDNA may become the primary screening tool with targeted swabbing for confirmation and detailed assessment.
Risk Modeling and Strategic Survey Design for BNG Wetlands
Geographic Risk Assessment
The Minnesota surveillance program developed a Bsal risk model and corresponding risk map shared with state natural resource agencies[1]. This approach stratifies landscapes based on:
- Host species distribution: Presence of susceptible amphibian populations
- Environmental suitability: Temperature and moisture regimes favorable to pathogen survival
- Introduction pathways: Proximity to pet trade routes, research facilities, or imported amphibians
- Connectivity: Hydrological links facilitating pathogen spread
BNG projects should consult regional risk assessments to calibrate survey intensity. High-risk sites warrant more comprehensive sampling, increased frequency, and enhanced biosecurity protocols.
Adaptive Survey Frameworks
Risk-stratified approach:
Low-risk sites:
- Baseline eDNA screening during initial habitat assessment
- Targeted swabbing if eDNA positive or clinical signs observed
- Annual monitoring during 30-year BNG management period
Moderate-risk sites:
- Combined eDNA and swab-based baseline survey
- Bi-annual monitoring for first 5 years
- Transition to annual surveillance if consistently negative
High-risk sites:
- Comprehensive swabbing of all detectable species
- Quarterly eDNA monitoring during establishment phase
- Immediate response protocols for positive detections
- Enhanced biosecurity for all site activities
This tiered framework optimizes resource allocation while maintaining surveillance coverage across BNG project portfolios. Small development projects with limited budgets can focus on baseline screening, while larger schemes implement intensive monitoring.
Integration with Biodiversity Metric Calculations
Chytrid detection status should influence habitat condition assessments within the statutory biodiversity metric. Infected wetlands represent compromised baseline conditions that may:
- Reduce calculated biodiversity units due to population stress
- Require intervention before habitat enhancement claims are valid
- Trigger revised management prescriptions to support disease resistance
The biodiversity impact assessment process must account for disease as a condition modifier, ensuring that unit calculations reflect genuine ecological functionality rather than superficially suitable habitat harboring compromised populations.
Biosecurity Protocols: Preventing Pathogen Introduction and Spread
Equipment Decontamination Standards
Chytrid zoospores survive on moist surfaces and equipment, making rigorous biosecurity essential for all wetland BNG activities:
Disinfection Protocol:
- Physical cleaning: Remove all visible soil and organic matter
- Disinfectant application: Virkon Aquatic (1% solution) or 10% bleach solution
- Contact time: Minimum 15 minutes for complete pathogen inactivation
- Rinse: Use clean water to remove disinfectant residues
- Drying: Allow complete air-drying before next use (zoospores are vulnerable to desiccation)
Equipment requiring decontamination:
- Waders and boots
- Nets and traps
- Survey equipment (measuring devices, cameras)
- Vehicles (tire treads, undercarriage)
- Construction machinery entering wetland zones
Site Movement Protocols
Movement between wetland BNG sites creates the highest transmission risk. Implement hierarchical site visitation:
Clean-to-dirty progression:
- Visit disease-free or unsampled sites first
- Progress to sites with unknown status
- End with confirmed positive sites (if applicable)
Between-site intervals:
- Minimum 24 hours between wetland visits after full decontamination
- Ideally 48 hours to ensure complete drying
- Use dedicated equipment for high-risk sites when possible
The 2024 Association of Fish and Wildlife Agencies guidance emphasizes early prevention and preparedness as the most cost-effective disease management strategy[1]. Biosecurity investment during project planning prevents far more expensive remediation efforts later.
Amphibian Translocation Considerations
BNG projects sometimes involve amphibian translocations to newly created habitats. These movements carry extreme disease risk and require:
✋ Pre-movement screening: All individuals must test negative for both Bd and Bsal
✋ Quarantine periods: Minimum 60 days with repeat testing before release
✋ Source population health: Document disease-free status of donor sites
✋ Veterinary oversight: Involve amphibian disease specialists in planning
✋ Regulatory compliance: Obtain necessary permits acknowledging disease considerations
Many ecologists now recommend habitat creation without translocation, allowing natural colonization to occur. This approach eliminates translocation-associated disease risks while supporting genetic connectivity through natural dispersal.
Responding to Positive Detections in BNG Projects
Immediate Actions
Positive chytrid detection triggers a structured response sequence:
Phase 1: Confirmation (0-2 weeks)
- Conduct follow-up sampling to confirm initial findings
- Expand survey area to define infection extent
- Document clinical disease signs in affected populations
- Notify relevant regulatory authorities and stakeholders
Phase 2: Assessment (2-6 weeks)
- Quantify infection prevalence and intensity
- Evaluate population-level impacts
- Assess transmission risk to adjacent habitats
- Consult amphibian disease specialists for guidance
Phase 3: Management (6 weeks onward)
- Implement enhanced biosecurity for all site activities
- Revise habitat management to support disease resistance
- Consider intervention options (if appropriate)
- Adjust biodiversity unit calculations to reflect condition
Management Options
Habitat manipulation:
- Increase habitat heterogeneity to provide thermal refugia
- Enhance terrestrial habitat quality to reduce aquatic dependency
- Create shallow areas that warm quickly (Bd is temperature-sensitive)
- Maintain water quality to support immune function
Population support:
- Reduce other stressors (predation, competition, pollution)
- Protect overwintering sites
- Maintain connectivity to facilitate genetic diversity
- Monitor for population recovery signals
Containment:
- Restrict equipment movement from infected sites
- Enhanced decontamination for all activities
- Limit hydrological connections where feasible
- Educate site users about transmission risks
Treatment considerations:
Direct antifungal treatment of wild amphibians remains experimental and rarely practical for free-living populations. Captive assurance colonies may be appropriate for critically endangered species, but most BNG contexts focus on habitat-based management and biosecurity.
Long-term Monitoring Adjustments
Detected infections require permanent surveillance protocol changes:
- Increase monitoring frequency to track disease dynamics
- Expand spatial coverage to detect spread
- Document population trends relative to infection status
- Contribute data to national disease surveillance networks
- Revise 30-year BNG management plans to incorporate disease considerations
Transparency about disease status protects all stakeholders. Developers seeking to achieve biodiversity net gain benefit from accurate baseline data that supports defensible unit calculations and realistic management expectations.
Integrating Chytrid Surveillance into BNG Compliance Frameworks
Regulatory Context in 2026
While current BNG legislation doesn't explicitly mandate chytrid surveillance, several regulatory drivers create implicit requirements:
Condition assessments: The statutory biodiversity metric requires accurate habitat condition scoring, which disease presence affects
30-year monitoring: Long-term management obligations necessitate understanding baseline disease status to interpret future population changes
Due diligence: Professional ecological consultants have duty-of-care obligations to identify significant threats to biodiversity outcomes
Biosecurity regulations: Existing wildlife disease control measures apply to BNG activities involving amphibian habitats
Forward-thinking biodiversity plans now incorporate disease surveillance as standard practice, recognizing that pathogen-free status represents genuine ecological value worth documenting and protecting.
Cost-Benefit Analysis for Developers
Survey investment:
- Baseline swab survey (30 samples): £1,500-£3,000
- eDNA screening (5 samples): £800-£1,500
- Annual monitoring: £500-£1,000
Avoided costs:
- Failed biodiversity unit delivery: £10,000-£100,000+
- Remediation and re-establishment: £50,000-£500,000+
- Regulatory enforcement: Variable
- Reputational damage: Difficult to quantify
The cost-benefit ratio strongly favors proactive surveillance, particularly for larger projects where biodiversity unit values are substantial. Off-site habitat banking schemes especially benefit from disease-free certification, as it enhances unit marketability and reduces long-term liability.
Documentation and Reporting Standards
Comprehensive chytrid surveillance generates evidence supporting BNG compliance:
Baseline reports should include:
- Survey methodology and timing
- Sample sizes and species representation
- Laboratory analysis protocols
- Results summary with spatial distribution
- Biosecurity measures implemented
- Risk assessment and management recommendations
Ongoing monitoring documentation:
- Annual surveillance results
- Population trend analysis
- Disease status changes
- Management interventions implemented
- Contribution to regional disease databases
This documentation demonstrates due diligence and provides defensible evidence that biodiversity outcomes reflect genuine ecological gains rather than disease-masked habitat quality.
Future Directions: Emerging Technologies and Adaptive Management
Technological Innovations
Next-generation sequencing: Metagenomic approaches can simultaneously detect multiple pathogens, parasites, and microbiome changes from single samples, providing comprehensive health assessments.
Portable PCR devices: Field-deployable molecular testing enables real-time results during surveys, facilitating immediate decision-making.
Remote sensing integration: Thermal imaging and multispectral analysis may eventually detect stressed amphibian populations before clinical disease becomes apparent.
Artificial intelligence: Machine learning algorithms can analyze disease distribution patterns and predict outbreak risks based on environmental variables.
Adaptive Management Frameworks
BNG projects operate over 30-year timescales, requiring flexible approaches that incorporate new knowledge:
- Annual protocol review: Update survey methods as technologies improve
- Risk reassessment: Revise geographic risk models as disease distributions change
- Management experimentation: Test habitat interventions and document outcomes
- Knowledge sharing: Contribute findings to collective understanding of disease ecology
The Minnesota surveillance program's partnership with over 14 organizations demonstrates the value of collaborative networks[1]. UK BNG practitioners should similarly engage with research institutions, conservation organizations, and regulatory agencies to build collective capacity for disease management.
Research Priorities
Critical knowledge gaps requiring investigation:
🔬 UK-specific pathogen ecology: How do Bd and potential Bsal behave in British wetland conditions?
🔬 Habitat resistance factors: What design elements support disease-resistant amphibian populations?
🔬 Intervention effectiveness: Which management actions successfully reduce infection impacts?
🔬 Climate interactions: How will changing temperatures affect pathogen-host dynamics?
🔬 Population recovery: What conditions enable amphibian populations to persist with endemic infections?
BNG projects represent opportunities for applied research that advances both practical management and scientific understanding. Landowners and developers who support research collaboration generate knowledge benefiting the entire conservation community.
Conclusion
Amphibian Chytrid Fungus Surveys in Wetland BNG Projects: Early Detection Strategies for Ecologists represents an essential evolution in biodiversity net gain practice for 2026 and beyond. As chytrid fungi continue threatening amphibian populations globally, UK wetland creation and enhancement projects must embed disease surveillance into standard protocols—not as optional add-ons, but as fundamental components of ecological due diligence.
The integration of swab-based detection and eDNA methods provides ecologists with complementary tools for establishing disease-free baselines and monitoring pathogen emergence. Risk-stratified survey frameworks optimize resource allocation while maintaining comprehensive coverage across project portfolios. Rigorous biosecurity protocols prevent the inadvertent introduction and spread of these devastating pathogens between sites.
For developers, the investment in chytrid surveillance protects biodiversity unit integrity and reduces long-term liability. For ecologists, systematic disease monitoring enhances professional standards and contributes to national conservation efforts. For amphibian populations, early detection creates intervention opportunities that may prevent local extinctions.
Actionable Next Steps
For ecologists planning BNG surveys:
- Incorporate chytrid screening into baseline amphibian assessments
- Adopt risk-stratified protocols based on site characteristics
- Implement comprehensive biosecurity for all wetland activities
- Establish relationships with qualified disease testing laboratories
- Contribute findings to regional disease surveillance networks
For developers and landowners:
- Request chytrid surveillance as standard component of biodiversity assessments
- Budget for baseline screening and ongoing monitoring
- Ensure contractors follow biosecurity protocols
- Document disease-free status to enhance biodiversity unit value
- Support long-term monitoring through 30-year management period
For regulatory and policy stakeholders:
- Consider explicit disease surveillance requirements in BNG guidance
- Support development of UK-specific risk mapping
- Facilitate data sharing through national disease portals
- Fund research on habitat-based disease management
- Recognize disease-free status in condition assessments
The chytrid fungus threat will not diminish—but through systematic surveillance, rigorous biosecurity, and adaptive management, BNG projects can protect both amphibian populations and biodiversity outcomes. Early detection remains the most powerful tool available, transforming a potential catastrophe into a manageable challenge.
The time to act is now, during the establishment phase of England's BNG framework. By embedding disease surveillance into standard practice from the outset, the ecological consulting community can ensure that wetland BNG projects deliver genuine, lasting biodiversity gains for decades to come.
References
[1] Chytridfungi – https://maisrc.umn.edu/research-projects/chytridfungi
[2] Pmc12789641 – https://pmc.ncbi.nlm.nih.gov/articles/PMC12789641/
[3] Usgs:5af5ef0de4b0da30c1b5f9eb – https://data.usgs.gov/datacatalog/data/USGS:5af5ef0de4b0da30c1b5f9eb


